

基于电化学发光的砷(III)传感器:采用丝网印刷金电极上的鲁米诺
收稿日期: 2025-07-09
修回日期: 2025-10-09
录用日期: 2025-11-14
网络出版日期: 2025-11-14
An Electrochemiluminescence-Based Arsenic (III) Sensor using Luminol on Screen-Printed Gold Electrodes
Received date: 2025-07-09
Revised date: 2025-10-09
Accepted date: 2025-11-14
Online published: 2025-11-14
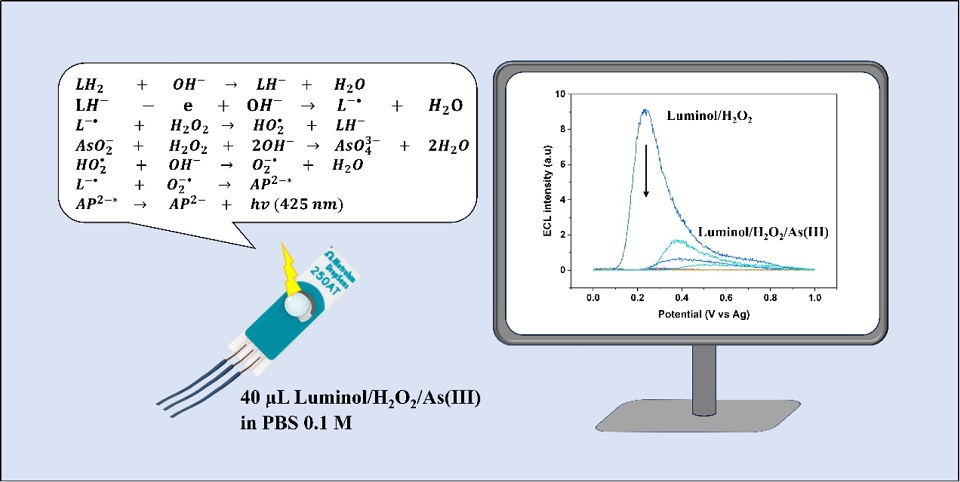
在丝网印刷金电极上研究了鲁米诺的电化学发光,用于简单而灵敏地检测砷离子(砷(III))。采用循环伏安法研究了鲁米诺的电化学行为,并评估了砷离子对电化学发光体系的影响;同时以过氧化氢作为共反应物,在碱性条件下增强了鲁米诺发光效率。通过精细优化pH值、扫描速率及过氧化氢/鲁米诺浓度等关键参数,实现了电极性能最优化。结果表明,砷(III)的存在对鲁米诺/过氧化氢电化学发光系统产生淬灭效应,导致该系统在1 nmol·L-1至150 µmol·L-1的宽浓度范围内电化学发光信号呈线性衰减,展现出1.21 nmol·L-1的低检测限,并具有2.27%相对标准偏差的优异重复性,彰显其在砷(III)检测中的灵敏度与可靠性。本研究的关键优势在于成功采用市售裸电极——这类电极易于获取且无需改性,充分验证了其在电化学发光砷检测中的有效性。优化后的pH 10缓冲液在提升砷检测选择性方面发挥关键作用:该条件既促进鲁米诺最佳去质子化反应,又确保砷保持溶解态,而其他潜在金属离子干扰物则更易形成固态金属(氢)氧化物。此外,本文研发的传感器还成功应用于海水基质中的砷(III)检测,证明其作为基于电化学发光技术的砷传感器在环境应用中具有可靠且高效的潜力。
哈梅萨·哈梅萨 , 伊斯娜伊妮·拉赫玛瓦蒂 , 安德烈亚·菲奥拉尼 , 永永康昭 , 恩妮·库斯里尼 , 阿安·约翰·瓦尤迪 , 阿塞普·赛富米拉 , 特里比达萨里·A·伊万迪尼 . 基于电化学发光的砷(III)传感器:采用丝网印刷金电极上的鲁米诺[J]. 电化学, 2026 , 32(2) : 2507092 . DOI: 10.61558/2993-074X.3593
Electrochemiluminescence (ECL) of luminol has been studied on a screen-printed gold electrode for a simple and sensitive detection of arsenic ions (As(III)). Cyclic voltammetry (CV) was applied as the proposed technique to study luminol's electrochemical behavior and to evaluate the arsenic’s effect in the ECL system, while hydrogen peroxide (H2O2) served as a co-reactant to enhance luminol’s light emission under alkaline conditions. To achieve optimal electrode performance, key parameters including pH, scan rate, and the concentrations of H2O2 and luminol were carefully optimized. The presence of As(III) induced a quenching effect on the luminol/H2O2 ECL system, leading to a linear decrease in ECL signal across the wide concentration range of 1 nmol·L-1 to 150 µmol·L-1. The system demonstrated a low detection limit of 1.21 nmol·L-1 and exhibited excellent repeatability with a relative standard deviation of 2.27%, highlighting its sensitivity and reliability for As(III) detection. A key advantage of this study was the successful use of commercial bare electrodes, which were readily available and required no modifications, proving their effectiveness for ECL-based arsenic sensing. The optimized buffer solution pH of 10 played a critical role in enhancing arsenic detection selectivity, as it facilitated the optimal deprotonation of luminol and ensured arsenic remained in its dissolved state, whereas other potential metal ion interferences were more likely to form solid metal (hydro)oxides. Furthermore, the developed sensor was successfully applied for As(III) detection in a seawater matrix, demonstrating its potential as a robust and effective ECL-based arsenic sensor for environmental applications.

| [1] | Amen R, Bashir H, Bibi I, Shaheen S M, Niazi N K, Shahid M, Hussain M M, Antoniadis V, Shakoor M B, Al-Solaimani S G, Wang H L, Bundschuh J, Rinklebe J. A critical review on arsenic removal from water using biochar-based sorbents: The significance of modification and redox reactions[J]. Chem. Eng. J., 2020, 396: 125195. https://doi.org/10.1016/j.cej.2020.125195. |
| [2] | kaur R, Garkal A, Sarode L, Bangar P, Mehta T, Singh D P, Rawal R. Understanding arsenic toxicity: Implications for environmental exposure and human health[J]. J. Hazard. Mater. Lett., 2024, 5: 100090. https://doi.org/10.1016/j.hazl.2023.100090. |
| [3] | Rahaman M S, Rahman M M, Mise N, Sikder M T, Ichihara G, Uddin M K, Kurasaki M, Ichihara S. Environmental arsenic exposure and its contribution to human diseases, toxicity mechanism and management[J]. Environ. Pollut., 2021, 289: 117940. https://doi.org/10.1016/j.envpol.2021.117940. |
| [4] | Sharma V K, Sohn M. Aquatic arsenic: Toxicity, speciation, transformations, and remediation[J]. Environ. Int., 2009, 35(4): 743-759. https://doi.org/10.1016/j.envint.2009.01.005. |
| [5] | Alka S, Shahir S, Ibrahim N, Ndejiko M J, Vo D V N, Manan F A. Arsenic removal technologies and future trends: A mini review[J]. J. Clean. Prod., 2021, 278: 123805. https://doi.org/10.1016/j.jclepro.2020.123805. |
| [6] | Sevak P, Pushkar B. Arsenic pollution cycle, toxicity and sustainable remediation technologies: A comprehensive review and bibliometric analysis[J]. J. Environ. Manage., 2024, 349: 119504. https://doi.org/10.1016/j.jenvman.2023.119504. |
| [7] | Raju N J. Arsenic in the geo-environment: A review of sources, geochemical processes, toxicity and removal technologies[J]. Environ. Res., 2022, 203: 111782. https://doi.org/10.1016/j.envres.2021.111782. |
| [8] | Aktar S, Mia S, Makino T, Rahman M M, Rajapaksha A U. Arsenic removal from aqueous solution: A comprehensive synthesis with meta-data[J]. Sci. Total Environ., 2023, 862: 160821. https://doi.org/10.1016/j.scitotenv.2022.160821. |
| [9] | Herath I, Kumarathilaka P, Bundschuh J, Marchuk A, Rinklebe J. A fast analytical protocol for simultaneous speciation of arsenic by ultra-high performance liquid chromatography (UHPLC) hyphenated to inductively coupled plasma mass spectrometry (ICP-MS) as a modern advancement in liquid chromatography approaches[J]. Talanta, 2020, 208: 120457. https://doi.org/10.1016/j.talanta.2019.120457. |
| [10] | Pétursdóttirá H, Gunnlaugsdóttir H. Selective and fast screening method for inorganic arsenic in seaweed using hydride generation inductively coupled plasma mass spectrometry (HG-ICPMS)[J]. Microchem. J., 2019, 144: 45-50. https://doi.org/10.1016/j.microc.2018.08.055. |
| [11] | Harmesa H, Wahyudi A J, Saefumillah A, Ivandini T A. Electrochemiluminescence systems for metal-ion detection: a systematic review[J]. ChemistrySelect, 2024, 9(21): 1-24. https://doi.org/10.1002/slct.202401544. |
| [12] | Ivandini T A, Sato R, Makide Y, Fujishima A, Einaga Y. Electrochemical detection of arsenic(III) using indium-implanted boron-doped diamond electrodes[J]. Anal. Chem., 2006, 78(18): 6291-6298. https://doi.org/10.1021/ac0519514. |
| [13] | Agustiany T, Khalil M, Einaga Y, Jiwanti P K, Ivandini T A. Stable iridium-modified boron-doped diamond electrode for the application in electrochemical detection of arsenic (III)[J]. Mater. Chem. Phys., 2020, 244: 1-7. https://doi.org/10.1016/j.matchemphys.2020.122723. |
| [14] | Rahmawati I, Fiorani A, Sanjaya A R, Irkham, Du J, Saepudin E, Einaga Y, Ivandini T A. Modification of boron-doped diamond electrode with polyaniline and gold particles to enhance the electrochemiluminescence of luminol for the detection of reactive oxygen species (hydrogen peroxide and hypochlorite)[J]. Diam. Relat. Mater., 2024, 144: 110956. https://doi.org/10.1016/j.diamond.2024.110956. |
| [15] | Rahmawati I, Saepudin E, Fiorani A, Einaga Y, Ivandini T A. Electrogenerated chemiluminescence of luminol at a boron-doped diamond electrode for the detection of hypochlorite[J]. Analyst, 2022, 147(12): 2696-2702. https://doi.org/10.1039/d2an00540a. |
| [16] | Wang Y R, Zhao X Y, Qu N R, Gu J M. Electrochemiluminescence for high-performance Chiral recognition of enantiomers: Recent advances and future perspectives[J]. Org. Electron., 2023, 122: 106902. https://doi.org/10.1016/j.orgel.2023.106902. |
| [17] | Husain R A, Barman S R, Chatterjee S, Khan I, Lin Z H. Enhanced biosensing strategies using electrogenerated chemiluminescence: recent progress and future prospects[J]. J. Mater. Chem., B. 2020, 8(16): 3192-3212. https://doi.org/10.1039/c9tb02578b. |
| [18] | Zanut A, Fiorani A, Canola S, Saito T, Ziebart N, Rapino S, Rebeccani S, Barbon A, Irie T, Josel H P, Negri F, Marcaccio M, Windfuhr M, Imai K, Valenti G, Paolucci F. Insights into the mechanism of coreactant electrochemiluminescence facilitating enhanced bioanalytical performance[J]. Nat. Commun., 2020, 11(1): 2668. https://doi.org/10.1038/s41467-020-16476-2. |
| [19] | Truong C K P, Nguyen T D D, Shin I S. Electrochemiluminescent chemosensors for clinical applications: a review[J]. Biochip J., 2019, 13(3): 203-216. https://doi.org/10.1007/s13206-019-3301-9. |
| [20] | Ying X D, Zhou L, Fu W X, Wang Y F, Su B. Electrochemiluminescence devices for point-of-care testing[J]. Sens. Diagn., 2023, 2(3): 480-491. https://doi.org/10.1039/d2sd00232a. |
| [21] | Hao N, Wang K. Recent development of electrochemiluminescence sensors for food analysis[J]. Anal. Bioanal. Chem., 2016, 408(25): 7035-7048. https://doi.org/10.1007/s00216-016-9548-2. |
| [22] | Zhou J J, Lv X Q, Jia J L, Din Z U, Cai S Q, He J L, Xie F, Cai J. Nanomaterials-based electrochemiluminescence biosensors for food analysis: recent developments and future directions[J]. Biosensors, 2022, 12(11): 1046. https://doi.org/10.3390/bios12111046. |
| [23] | Wang X B, Zhao Y Q, Hua Q, Lu J J, Tang F Y, Sun W J, Luan F, Zhuang X M, Tian C Y. An ultrasensitive electrochemiluminescence biosensor for the detection of total bacterial count in environmental and biological samples based on a novel sulfur quantum dot luminophore[J]. Analyst, 2022, 147(8): 1716-1721. https://doi.org/10.1039/d2an00153e. |
| [24] | Li L L, Che Y, Zhu J J. Recent advances in electrochemiluminescence analysis[J]. Anal. Chem., 2017, 89(1): 358-371. https://doi.org/10.1021/acs.analchem.6b04675. |
| [25] | Shen Y, Gao X, Lu H J, Nie C, Wang J L. Electrochemiluminescence-based innovative sensors for monitoring the residual levels of heavy metal ions in environment-related matrices[J]. Coord. Chem. Rev., 2023, 476: 214927. https://doi.org/10.1016/j.ccr.2022.214927. |
| [26] | Liang R P, Yu L D, Tong Y J, Wen S H, Cao S P, Qiu J D. An ultratrace assay of arsenite based on the synergistic quenching effect of Ru(bpy)32+ and arsenite on the electrochemiluminescence of Au-g-C3N4 nanosheets[J]. Chem. Commun., 2018, 54(99): 14001-14004. https://doi.org/10.1039/c8cc08353c. |
| [27] | Wang R Z, Zhao Y, Jie G F. A novel DNA-quantum dot nanostructure electrochemiluminescence aptamer sensor by chain reaction amplification for rapid detection of trace Cd2+[J]. Analyst, 2023, 148(19): 4844-4849. https://doi.org/10.1039/d3an01247f. |
| [28] | Huang R F, Liu H X, Gai Q Q, Liu G J, Wei Z. A facile and sensitive electrochemiluminescence biosensor for Hg2+ analysis based on a dual-function oligonucleotide probe[J]. Biosens. Bioelectron., 2015, 71: 194-199. https://doi.org/10.1016/j.bios.2015.04.038. |
| [29] | Zhao G H, Li X J, Zhao Y B, Li Y Y, Cao W, Wei Q. Electrochemiluminescence assay of Cu2+by using one-step electrodeposition synthesized CdS/ZnS quantum dots[J]. Analyst, 2017, 142(17): 3272-3277. https://doi.org/10.1039/c7an01014a. |
| [30] | Zhang Y, Xu J M, Zhou S, Zhu L, Lv X, Zhang J, Zhang L, Zhu P H, Yu J H. DNAzyme-triggered visual and ratiometric electrochemiluminescence dual-readout assay for Pb(II) based on an assembled paper device[J]. Anal. Chem., 2020, 92(5): 3874-3881. https://doi.org/10.1021/acs.analchem.9b05343. |
| [31] | Zakaria N D, Salih I L, Hamzah H H, S?nmez T, Omar M H, Nor N M, Razak K A, Balakrishnan V. Electrochemical and imaging evaluations of electrochemically activated screen-printed gold electrodes[J]. Analyst, 2024, 149(22): 5401-5410. https://doi.org/10.1039/d4an00990h. |
| [32] | Kozak J, Tyszczuk-Rotko K. Screen-printed gold electrode for ultrasensitive voltammetric determination of the antipsychotic drug thioridazine[J]. Meas. J. Int. Meas. Confed., 2023, 217: 113107. https://doi.org/10.1016/j.measurement.2023.113107. |
| [33] | Liu Z G, Huang X J. Voltammetric determination of inorganic arsenic[J]. TrAC - Trends Anal. Chem., 2014, 60: 25-35. https://doi.org/10.1016/j.trac.2014.04.014. |
| [34] | Duan Y, Song Y, Fan N K, Yao Y Z, Deng S X, Ding S J, Shen B, Yin Q F. Self-enhanced luminol-based electrochemiluminescent hydrogels: An ultrasensitive biosensing platform for fusion gene analysis coupled with target-initiated DNAzyme motor[J]. Biosens. Bioelectron., 2022, 197: 113784. https://doi.org/10.1016/j.bios.2021.113784. |
| [35] | Liu G P, Yuan Y L, Wang J L. Hemin/G-quadruplex DNAzyme nanowires amplified luminol electrochemiluminescence system and its application in sensing silver ions[J]. RSC Adv., 2016, 6(43): 37221-37225. https://doi.org/10.1039/c6ra01809b. |
| [36] | Hu Y X, Liu Y, Wang S, Guo Z Y, Hu Y F, Xie H Z. A novel surface-tethered double-signal electrochemiluminescence sensor based on luminol@Au and Cds quantum dots for mercury ion detection[J]. ChemistrySelect, 2019, 4(10): 2926-2932. https://doi.org/10.1002/slct.201802150. |
| [37] | Zhao C L, Ma C Y, Zhang F P, Li W J, Hong C L, Qi Y. Two-dimensional metal-organic framework nanosheets: An efficient two-electron oxygen reduction reaction electrocatalyst for boosting cathodic luminol electrochemiluminescence[J]. Chem. Eng. J., 2023, 466: 1-8. https://doi.org/10.1016/j.cej.2023.143156. |
| [38] | Huang Y T, Zhang S P, Chen S S, Chen Y J, Cheng L J, Dai H, Gao L H. Electrochemiluminescence enhanced by molecular engineering linear π-conjugated polymer: An ingenious ECL emitter for the construction of exosome sensing platform[J]. Talanta, 2024, 277: 126405. https://doi.org/10.1016/j.talanta.2024.126405. |
| [39] | Irkham, Rais R R, Ivandini T A, Fiorani A, Einaga Y. Electrogenerated chemiluminescence of luminol mediated by carbonate electrochemical oxidation at a boron-doped diamond, Anal. Chem., 2021, 93(4): 2336-2341. https://doi.org/10.1021/acs.analchem.0c04212. |
| [40] | Scholz F, Kahlert H. The calculation of the solubility of metal hydroxides, oxide-hydroxides, and oxides, and their visualisation in logarithmic diagrams[J]. ChemTexts, 2015, 1(1): 7. https://doi.org/10.1007/s40828-015-0006-0. |
| [41] | Ivandini T A, Yamada D, Watanabe T, Matsuura H, Nakano N, Fujishima A, Einaga Y. Development of amperometric arsine gas sensor using gold-modified diamond electrodes[J]. J. Electroanal. Chem., 2010, 645(1): 58-63. https://doi.org/10.1016/j.jelechem.2010.04.012. |
| [42] | Eikelboom M, Wang Y, Portlock G, Gourain A, Gardner J, Bullen J, Lewtas P, Carriere M, Alvarez A, Kumar A, O’Prey S, T?lgyes T, Omanovi? D, Bhowmick S, Weiss D, Salaun P. Voltammetric determination of inorganic arsenic in mildly acidified (pH 4.7) groundwaters from Mexico and India[J]. Anal. Chim. Acta., 2023, 1276: 341589. https://doi.org/10.1016/j.aca.2023.341589. |
| [43] | Giancarla A, Zanoni C, Merli D, Magnaghi L R, Biesuz R. A new cysteamine-copper chemically modified screen-printed gold electrode for glyphosate determination[J]. Talanta, 2024, 269: 125436. https://doi.org/10.1016/j.talanta.2023.125436. |
| [44] | Steel L, Ward A C, Jeffrey C, Alcorn D, Corrigan D K. Towards simple, rapid point of care testing for clinically important protein biomarkers of sepsis[J]. SCIOL Biotechnol., 2017, 1: 1-8. https://doi.org/10.11648/j.sjbt.20170101.11. |
| [45] | Mccormick W J, Doran W J. Covalent immobilisation of a nanoporous platinum film onto a gold screen-printed electrode for highly stable and selective[J]. Catalysts, 2021, 11(10): 1161. https://doi.org/10.3390/catal11101161. |
| [46] | Monnappa A B, Manjunatha J G, Bhatt A S. Design of a sensitive and selective voltammetric sensor based on a cationic surfactant-modified carbon paste electrode for the determination of alloxan[J]. ACS Omega, 2020, 5(36): 23481-23490. https://doi.org/10.1021/acsomega.0c03517. |
| [47] | Dhaffouli A, Moussaoui Y, Salazar-Carballo P A, Holzinger M, Carinelli S, Barhoumi H. Design of a high-performance electrochemical sensor using ZnO@SiO?-n-propyl-NH?-benzothiazole-AuNPs for the selective and sensitive detection of lead ions in environmental samples[J]. J. Electrochem. Soc., 2025, 172(6): 067520. https://doi.org/10.1149/1945-7111/ade47f. |
| [48] | Yin F, Sun Q, Huang X Z, Wu G Q, Zhang Y J, Shen Y F. Recent progress in signal enhancement of nanomaterials-based electrochemiluminescence systems[J]. TrAC-Trends Anal. Chem., 2023, 169: 117376. https://doi.org/10.1016/j.trac.2023.117376. |
| [49] | Zhou P, Hu S J, Guo W L, Su B. Deciphering electrochemiluminescence generation from luminol and hydrogen peroxide by imaging light emitting layer[J]. Fundam. Res., 2022, 2(5): 682-687. https://doi.org/10.1016/j.fmre.2021.11.018. |
| [50] | Pettine M, Campanella L, Millero F J. Arsenite oxidation by H2O2 in aqueous solution[J]. Geochim. Cosmochim. Acta, 1999, 63(18): 2727-2735. https://doi.org/10.1139/v01-027. |
| [51] | Molnár L, Vir?íkova E, Lech P. Experimental study of As(III) oxidation by hydrogen peroxide[J]. Hydrometallurgy, 1994, 35(1): 1-9. https://doi.org/10.1016/0304-386X(94)90013-2. |
| [52] | Pourbaix M, Zhang H, Pourbaix A. Presentation of an atlas of chemical and electrochemical equilibria in the presence of a gaseous phase[J]. Mater. Sci. Forum., 1997, 251-252: 143-148. https://doi.org/10.4028/www.scientific.net/msf.251-254.143. |
| [53] | Liu Y, Hu Y X, Wang S, Guo Z Y, Hu Y F. A novel surface-tethered analysis method for mercury (II) ion detection via self-assembly of individual electrochemiluminescence signal units[J]. Electroanalysis, 2018, 30(5): 859-867. https://doi.org/10.1002/elan.201700660. |
| [54] | Guo Z Y, Chen B B, Wang Z B, Jiang X H. An electrochemiluminescence biosensor for mercury ion detection based on gamma-polyglutamic acid-graphene-luminol composite and oligonucleotides[J]. Sens. Actuators B: Chem., 2015, 209: 579-585. https://doi.org/10.1016/j.snb.2014.12.028. |
| [55] | Xu M, Zhang Y, Li L, Kong Q K, Zhang L N, Ge S G, Yu J H. Colorimetric and electrochemiluminescence dual-mode sensing of lead ion based on integrated lab-on-paper device[J]. ACS Appl. Mater. Interfaces, 2018, 10(4): 3431-3440. https://doi.org/10.1021/acsami.7b18542. |
| [56] | Yildiz G, Tasdoven U, Menek N. Electrochemical characterization of luminol and its determination in real samples[J]. Anal. Methods, 2014, 6(19): 7809-7813. https://doi.org/10.1039/c4ay01281j. |
| [57] | Rajendran S, Ramanaiah D V, Kundu S, Bhunia S K. Yellow fluorescent carbon dots for selective recognition of As3+ and Fe3+ ions[J]. ACS Appl. Nano Mater., 2021, 4(10): 10931-10942. https://doi.org/10.1021/acsanm.1c02391. |
| [58] | Sepúlveda A C, Gatti L M, Kerl C F, Chennu A, Klatt J M. Arsenic speciation analysis in porewater by a novel colorimetric assay[J]. Sci. Total Environ., 2022, 827: 154155. https://doi.org/10.1016/j.scitotenv.2022.154155. |
| [59] | Cui L, Wu J, Ju H X. Label-free signal-on aptasensor for sensitive electrochemical detection of arsenite[J]. Biosens. Bioelectron., 2016, 79: 861-865. https://doi.org/10.1016/j.bios.2016.01.010. |
| [60] | Zhang Y F, Li D L, Compton R G. Arsenic (III) detection with underpotential deposition on gold[J]. J. Electroanal. Chem., 2022, 909: 116154. https://doi.org/10.1016/j.jelechem.2022.116154. |
| [61] | Kaimal R, Maridevaru M C, Dube A, Wu J J, Sambandam A, Ashokkumar M. Borophene nanosheet-based electrochemical sensing toward groundwater arsenic detection[J]. Ind. Eng. Chem. Res., 2023, 62(38): 15418-15427. https://doi.org/10.1021/acs.iecr.3c01922. |
/
| 〈 |
|
〉 |