

轴向硫配位工程增强Fe‒N‒C催化剂性能,助力高性能质子交换膜燃料电池
收稿日期: 2025-09-28
修回日期: 2025-10-27
录用日期: 2025-11-05
网络出版日期: 2025-11-05
Axial Sulfur-Coordination Engineering Boosting Fe‒N‒C Catalysts for High-Performance Proton Exchange Membrane Fuel Cells
Contributed equally to this work as the co-first authors.
Received date: 2025-09-28
Revised date: 2025-10-27
Accepted date: 2025-11-05
Online published: 2025-11-05
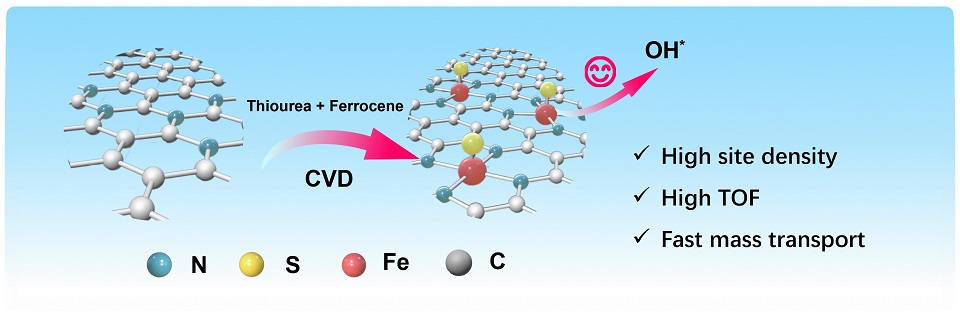
Fe-N-C催化剂长期受限于氧还原反应动力学迟缓的问题,这源于其对氧中间体的过度吸附强度及较低的活性位点利用率。杂原子掺杂通过调节金属位点的电子结构优化中间体吸附,有效加速氧还原反应动力学,而化学气相沉积技术可以提升活性位点的周转频率。本文采用双前驱体的化学气相沉积策略,开发出富含FeS1N4位点的FeSNC催化剂。实验与理论分析表明,硫元素的引入打破了活性位点的对称配位,将OH*吸附能从0.212 eV优化至1.194 eV。此外,活性位点的周转频率从1.98 e-1·site-1·s-1提升至6.32 e-1·site-1·s-1,显著增强了催化剂的本征活性。更值得注意的是,含硫物种的亲水性显著提高了掺硫催化剂的亲水性,从而促进了氧气和质子传输。因此,FeSNC催化剂在0.1 mol·L-1 HClO4中表现出极高的0.863 V半波电位,并在H2-O2质子交换膜燃料电池中达到1.2 W·cm-2的峰值功率密度。这项工作突出了配位工程的关键作用。
林琳 , 候秀旋 , 樊哲琛 , 尹义轩 , 赵纬祎 , 魏恺 , 周雨蝶 , 候利娜 , 王颖 , 万浩 , 葛君杰 . 轴向硫配位工程增强Fe‒N‒C催化剂性能,助力高性能质子交换膜燃料电池[J]. 电化学, 2026 , 32(3) : 2509281 . DOI: 10.61558/2993-074X.3592
Fe-N-C catalysts have long suffered from kinetically sluggish oxygen reduction reaction (ORR) due to excessive adsorption strength toward oxygen intermediates and low site utilization. Heteroatom doping effectively accelerates ORR reaction kinetics through electronic structure modulation of metal sites for optimal intermediate adsorption, while chemical vapor deposition (CVD) enhances the turnover frequency (TOF) of active sites. Herein, we developed an FeSNC catalyst featuring abundant FeS1N4 sites via a dual-precursor CVD strategy. Experimental and theoretical analyses revealed that S incorporation disrupts the symmetric coordination of active sites, which optimizes OH* adsorption energies from 0.212 eV to 1.194 eV. Moreover, the TOF increased from 1.98 e-1·site-1·s-1 to 6.32 e-1·site-1·s-1, significantly enhancing the intrinsic activity of the catalyst. More notably, the hydrophilic character of S-containing species substantially improved hydrophilicity in the S-doped catalyst, thereby promoting mass transport of oxygen and proton delivery. As a result, the FeSNC catalyst exhibited an extremely high half-wave potential of 0.863 V in 0.1 mol·L-1 HClO4 and achieved a peak power density of 1.2 W·cm-2 in H2-O2 PEMFCs. This work highlights the critical role of coordination engineering.

| [1] | Jiao K, Xuan J, Du Q, Bao Z M, Xie B, Wang B W, Zhao Y, Fan L H, Wang H Z, Hou Z J, Huo S, Brandon N P, Yin Y, Guiver M D. Designing the next generation of proton-exchange membrane fuel cells[J]. Nature, 2021. 595(7867): 361-369. http://dx.doi.org/10.1038/s41586-021-03482-7. |
| [2] | Scofield M E, Liu H, Wong S S. A concise guide to sustainable PEMFCs: recent advances in improving both oxygen reduction catalysts and proton exchange membranes[J]. Chem. Soc. Rev., 2015, 44(16): 5836-5860. http://dx.doi.org/10.1039/C5CS00302D. |
| [3] | Sun K, Dong J C, Sun H, Wang X D, Fang J J, Zhuang Z B, Tian S B, Sun X M. Co(CN)3 catalysts with well-defined coordination structure for the oxygen reduction reaction[J]. Nat. Catal., 2023, 6(12): 1164-1173. http://dx.doi.org/10.1038/s41929-023-01047-7. |
| [4] | Shao M H, Chang Q W, Dodelet J P, Chenitz R. Recent advances in electrocatalysts for oxygen reduction reaction[J]. Chem. Rev., 2016, 116(6): 3594-3657. http://dx.doi.org/10.1021/acs.chemrev.5b00462. |
| [5] | Zhao Y S, Wan J W, Yao H Y, Zhang L J, Lin K F, Wang L, Yang N L, Liu D B, Song L, Zhu J, Gu L, Liu L, Zhao H J, Li Y L, Wang D. Few-layer graphdiyne doped with sp-hybridized nitrogen atoms at acetylenic sites for oxygen reduction electrocatalysis[J]. Nat. Chem., 2018, 10(9): 924-931. http://dx.doi.org/10.1038/s41557-018-0100-1. |
| [6] | Snitkoff-Sol R Z, Rimon O, Bond A M, Elbaz L. Direct measurement of the oxygen reduction reaction kinetics on iron phthalocyanine using advanced transient voltammetry[J]. Nat. Catal., 2024, 7: 139-147. http://dx.doi.org/10.1038/s41929-023-01086-0. |
| [7] | Sa Y J, Seo D J, Woo J, Lim J T, Cheon J Y, Yang S Y, Lee J M, Kang D, Shin T J, Shin H S, Jeong H Y, Kim C S, Kim M G, Kim T Y, Joo S H. A general approach to preferential formation of active Fe-Nx sites in Fe-N/C electrocatalysts for efficient oxygen reduction reaction[J]. J. Am. Chem. Soc., 2016, 138(45): 15046-15056. http://dx.doi.org/10.1021/jacs.6b09470. |
| [8] | Zeng Y Z, Li C Z, Li B Y, Liang J S, Zachman M J, Cullen D A, Hermann R P, Alp E E, Lavina B, Karakalos S, Lucero M, Zhang B, Wang M, Feng Z X, Wang G F, Xie J, Myers D J, Dodelet J P, Wu G. Tuning the thermal activation atmosphere breaks the activity-stability trade-off of Fe-N-C oxygen reduction fuel cell catalysts[J]. Nat. Catal., 2023, 6(12): 1215-1227. http://dx.doi.org/10.1038/s41929-023-01062-8. |
| [9] | Gewirth A A, Varnell J A, DiAscro A M. Nonprecious metal catalysts for oxygen reduction in heterogeneous aqueous systems[J]. Chem. Rev., 2018, 118(5): 2313-2339. http://dx.doi.org/10.1021/acs.chemrev.7b00335. |
| [10] | Huang Z, Li F H, Liu Y D, Chen S G, Wei Z D, Tang Q. The role of nitrogen sources and hydrogen adsorption on the dynamic stability of Fe-N-C catalysts in oxygen reduction reaction[J]. Chem. Sci., 2024, 15(3): 1132-1142. http://dx.doi.org/https://doi.org/10.1039/d3sc05378d. |
| [11] | Xue D P, Yuan P F, Jiang S, Wei Y F, Zhou Y, Dong C L, Yan W F, Mu S, Zhang J N. Altering the spin state of Fe-N-C through ligand field modulation of single-atom sites boosts the oxygen reduction reaction[J]. Nano Energy, 2023, 105: 108020. http://dx.doi.org/https://doi.org/10.1016/j.nanoen.2022.108020. |
| [12] | Sui R, Liu B, Chen C, Tan X, He C, Xin D Y, Chen B, Xu Z Y, Li J Z, Chen W X, Zhuang Z B, Wang Z B, Chen C. Constructing asymmetric Fe-Nb diatomic sites to enhance ORR activity and durability[J]. J. Am. Chem. Soc., 2024, 146(38): 26442-26453. http://dx.doi.org/10.1021/jacs.4c09642. |
| [13] | Xie H, Xie X H, Hu G X, Prabhakaran V, Saha S, Gonzalez-Lopez L, Phakatkar A H, Hong M, Wu M L, Shahbazian-Yassar R, Ramani V, Al-Sheikhly M I, Jiang D E, Shao Y Y, Hu L B. Ta-TiOx nanoparticles as radical scavengers to improve the durability of Fe-N-C oxygen reduction catalysts[J]. Nat. Energy, 2022, 7(3): 281-289. http://dx.doi.org/10.1038/s41560-022-00988-w. |
| [14] | Li J Z, Zhang H G, Samarakoon W, Shan W T, Cullen D A, Karakalos S, Chen M L, Gu D M, More K L, Wang G, Feng Z, Wang Z, Wu G. Thermally driven structure and performance evolution of atomically dispersed FeN4 sites for oxygen reduction[J]. Angew. Chem. Int. Ed., 2019, 58(52): 18971-18980. http://dx.doi.org/https://doi.org/10.1002/anie.201909312. |
| [15] | Liu H, Jiang L Z, Khan J, Wang X X, Xiao J M, Zhang H D, Xie H J, Li L, Wang S Y, Han L. Decorating single-atomic Mn sites with FeMn clusters to boost oxygen reduction reaction[J]. Angew. Chem. Int. Ed., 2023, 62(3): e202214988. http://dx.doi.org/https://doi.org/10.1002/anie.202214988. |
| [16] | Yang N, Li L, Li J, Ding W, Wei Z D. Modulating the oxygen reduction activity of heteroatom-doped carbon catalysts via the triple effect: charge, spin density and ligand effect[J]. Chem. Sci., 2018, 9(26): 5795-5804. http://dx.doi.org/10.1039/C8SC01801D. |
| [17] | Wu S Q, Wei W, Li A Y. Theoretical studies on the bonding and separation performance of lanthanides and actinides with phenanthroline derived N,O/S-coordinated asymmetric hydrophilic ligands[J]. Comput. Theor. Chem., 2025, 1251: 115321. http://dx.doi.org/https://doi.org/10.1016/j.comptc.2025.115321. |
| [18] | Fan H Y, Liu Y R, Li J X, Lv Z H, Wang C L, Liu R, Dong F, Tian C G, FengX, Yang W X, Wang B. Optimizing the mass transport and atomic fe intrinsic activity to achieve high-performing fuel cells[J]. J. Am. Chem. Soc., 2025, 147(22): 18953-18962. http://dx.doi.org/10.1021/jacs.5c03499. |
| [19] | Ni W P, Gao Y, Zhang Y, Younus H A, Guo X, Ma C, Zhang Y, Duan J F, Zhang J, Zhang S G. O-doping boosts the electrochemical oxygen reduction activity of a single Fe site in hydrophilic carbon with deep mesopores[J]. ACS Appl. Mater. Interfaces, 2019, 11(49): 45825-45831. http://dx.doi.org/10.1021/acsami.9b18510. |
| [20] | Mehmood A, Gong M, Jaouen F, Roy A, Zitolo A, Khan A, Sougrati M T, Primbs M, Bonastre A M, Fongalland D, Drazic G, Strasser P, Kucernak A. High loading of single atomic iron sites in Fe-NC oxygen reduction catalysts for proton exchange membrane fuel cells[J]. Nat. Catal., 2022, 5: 311-323. http://dx.doi.org/10.1038/s41929-022-00772-9. |
| [21] | Kresse G, Furthmüller J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set[J]. Comput. Mater. Sci., 1996, 6(1): 15-50. http://dx.doi.org/https://doi.org/10.1016/0927-0256(96)00008-0. |
| [22] | Kresse G, Hafner J. Ab initio molecular dynamics for liquid metals[J]. Phys. Rev. B, 1993, 47: 558-561. http://dx.doi.org/10.1103/PhysRevB.47.558. |
| [23] | Blöchl P E. Projector augmented-wave method[J]. Phys. Rev. B, 1994, 50: 17953-17979. http://dx.doi.org/10.1103/PhysRevB.50.17953. |
| [24] | Perdew J P, Burke K, Ernzerhof M. Generalized gradient approximation made simple[J]. Phys. Rev. Lett., 1996, 77: 3865-3868. http://dx.doi.org/10.1103/PhysRevLett.77.3865. |
| [25] | Dudarev S L, Botton G A, Savrasov S Y, Humphreys C J, Sutton A P. Electron-energy-loss spectra and the structural stability of nickel oxide: An LSDA+U study[J]. Phys. Rev. B, 1998, 57: 1505-1509. http://dx.doi.org/10.1103/PhysRevB.57.1505. |
| [26] | Jiao Y C, Han D D, Liu L, Ji L, Guo G N, Hu J H, Yang D, Dong A G. Highly ordered mesoporous few-layer graphene frameworks enabled by Fe3O4 nanocrystal superlattices[J]. Angew. Chem. Int. Ed., 2015, 54(19): 5727-5731. http://dx.doi.org/https://doi.org/10.1002/anie.201501398. |
| [27] | Shen H J, Gracia-Espino E, Ma J Y, Tang H D, Mamat X, Wagberg T, Hu G Z, Guo S J. Atomically FeN2 moieties dispersed on mesoporous carbon: A new atomic catalyst for efficient oxygen reduction catalysis[J]. Nano Energy, 2017, 35: 9-16. http://dx.doi.org/https://doi.org/10.1016/j.nanoen.2017.03.027. |
| [28] | Wan-Me Z X, Zhang H T, Zhao Y, Wen H Z, Wan X Y, Xie Y L. Synthesis of S-doped mesoporous carbon and its use in advanced supercapacitors and sodium-ion batteries[J]. Carbon Trends, 2025, 21: 100558. http://dx.doi.org/https://doi.org/10.1016/j.cartre.2025.100558. |
| [29] | Shen H J, Gracia-Espino E, Ma J, Zang K T, Luo J, Wang L, Gao S S, Mamat X, Hu G, Wagberg T, Guo S J. Synergistic effects between atomically dispersed Fe-N-C and C-S-C for the oxygen reduction reaction in acidic media[J]. Angew. Chem. Int. Ed., 2017, 56(44): 13800-13804. http://dx.doi.org/https://doi.org/10.1002/anie.201706602. |
| [30] | Liu S W, Li C Z, Zachman M J, Zeng Y Z, Yu H R, Li B Y, Wang M Y, Braaten J, Liu J W, Meyer H M, Lucero M, Kropf A J, Alp E E, Gong Q, Shi Q, Feng Z X, Xu H, Wang G, Myers D J, Xie J, Cullen D A, Litster S, Wu G. Atomically dispersed iron sites with a nitrogen-carbon coating as highly active and durable oxygen reduction catalysts for fuel cells[J]. Nat. Energy, 2022, 7: 652-663. http://dx.doi.org/10.1038/s41560-022-01062-1. |
| [31] | Jia Q Y, Ramaswamy N, Tylus U, Strickland K, Li J, Serov A, Artyushkova K, Atanassov P, Anibal J, Gumeci C, Barton S C, Sougrati M T, Jaouen F, Halevi B, Mukerjee S. Spectroscopic insights into the nature of active sites in iron-nitrogen-carbon electrocatalysts for oxygen reduction in acid[J]. Nano Energy, 2016, 29: 65-82. http://dx.doi.org/https://doi.org/10.1016/j.nanoen.2016.03.025. |
| [32] | Jin Z Y, Jiao D X, Dong Y L, Liu L, Fan J, Gong M, Ma X C, Wang Y, Zhang W, Zhang L, Yu G Z, Voiry D, Zheng W T, Cui X Q. Boosting electrocatalytic carbon dioxide reduction via self-relaxation of asymmetric coordination in fe-based single atom catalyst[J]. Angew. Chem. Int. Ed., 2024, 136(6): e202318246. http://dx.doi.org/https://doi.org/10.1002/anie.202318246. |
| [33] | Zhao Y L, Chen H C, Ma X, Li J Y, Yuan Q, Zhang P, Wang M M, Li J, Li M, Wang S F, Guo H, Hu R, Tu K H, Zhu W, Li X N, Yang X, Pan Y. Vacancy defects inductive effect of asymmetrically coordinated single-atom Fe-N3S1 active sites for robust electrocatalytic oxygen reduction with high turnover frequency and mass activity[J]. Adv. Mater., 2024, 36(11): 2308243. http://dx.doi.org/https://doi.org/10.1002/adma.202308243. |
| [34] | Zhao L L, Cao Q, Wang A L, Duan J, Zhou W J, Sang Y H, Liu H. Iron oxide embedded titania nanowires - An active and stable electrocatalyst for oxygen evolution in acidic media[J]. Nano Energy, 2018, 45: 118-126. http://dx.doi.org/https://doi.org/10.1016/j.nanoen.2017.12.029. |
| [35] | Liang Y Y, Li Y G, Wang H L, Zhou J, Wang J, Regier T, Dai H J. Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction[J]. Nat. Mater., 2011, 10(10): 780-786. http://dx.doi.org/10.1038/nmat3087. |
| [36] | Wang W Y, Ren H, Guo J H, Wang S, Wang J, Wang Y Y, Sun W, Tang L, Wang X Q. Synergistically enhanced atomically dispersed Fe-Nx sites on carbon nanotubes by heteroatom S to boost electrochemical oxygen reduction for microbial fuel cell[J]. J. Power Sources, 2025, 629: 235960. http://dx.doi.org/https://doi.org/10.1016/j.jpowsour.2024.235960. |
| [37] | Han H, Wang X M, Zhang X. A hierarchically ordered porous Fe, N, S tri-doped carbon electrocatalyst with densely accessible Fe-Nx active sites and uniform sulfur-doping for efficient oxygen reduction reaction[J]. J. Colloid Interface Sci., 2022, 615: 617-626. http://dx.doi.org/https://doi.org/10.1016/j.jcis.2022.02.030. |
| [38] | Wang X, Chen W X, Zhang L, Yao T, Liu W, Lin Y, Ju H, Dong J C, Zheng L R, Yan W, Zheng X S, Li Z, Wang X Q, Yang J, He D S, Wang Y, Deng Z X, Wu Y, Li Y D. Uncoordinated amine groups of metal-organic frameworks to anchor single Ru sites as chemoselective catalysts toward the hydrogenation of quinoline[J]. J. Am. Chem. Soc., 2017, 139(28): 9419-9422. http://dx.doi.org/10.1021/jacs.7b01686. |
| [39] | Malko D, Kucernak A, Lopes T. In situ electrochemical quantification of active sites in Fe-N/C non-precious metal catalysts[J]. Nat. Commun., 2016, 7: 13285. http://dx.doi.org/10.1038/ncomms13285. |
| [40] | Liu Y R, Li J X, Lv Z H, Fan H, Dong F L, Wang C L, Chen X C, Liu R, Tian C A, Feng X, Yang W X, Wang B. Efficient proton-exchange membrane fuel cell performance of atomic Fe sites via p-d hybridization with Al dopants[J]. J. Am. Chem. Soc., 2024, 146(18): 12636-12644. http://dx.doi.org/10.1021/jacs.4c01598. |
| [41] | Nonoyama N, Okazaki S, Weber A Z, Ikogi Y, Yoshida T. Analysis of oxygen-transport diffusion resistance in proton-exchange-membrane fuel cells[J]. J. Electrochem. Sci., 2011, 158(4): B416-B423. http://dx.doi.org/10.1149/1.3546038. |
| [42] | Yin S H, Chen L, Yang J, Cheng X Y, Zeng H B, Hong Y H, Huang H, Kuai X X, Lin Y G, Huang R, Jiang Y X, Sun S G. A Fe-NC electrocatalyst boosted by trace bromide ions with high performance in proton exchange membrane fuel cells[J]. Nat. Commun., 2024, 15: 7489. http://dx.doi.org/10.1038/s41467-024-51858-w. |
| [43] | Yang B L, Han Q, Han L K, Leng Y, O'Carroll T, Yang X X, Wu G, Xiang Z H. Porous covalent organic polymer coordinated single Co site nanofibers for efficient oxygen-reduction cathodes in polymer electrolyte fuel cells[J]. Adv. Mater., 2023, 35(1): 2208661. http://dx.doi.org/https://doi.org/10.1002/adma.202208661. |
/
| 〈 |
|
〉 |