王文琪, 金杰, 汪丽敏, 刘馨月, 程涛, 厚镛, 薛涵, 王志宇, 刘博, 刘佳保, 路旭斌
. 全钒液流电池电极材料研究进展[J]. 电化学, 0
: 0
.
DOI: 10.61558/2993-074X.3589
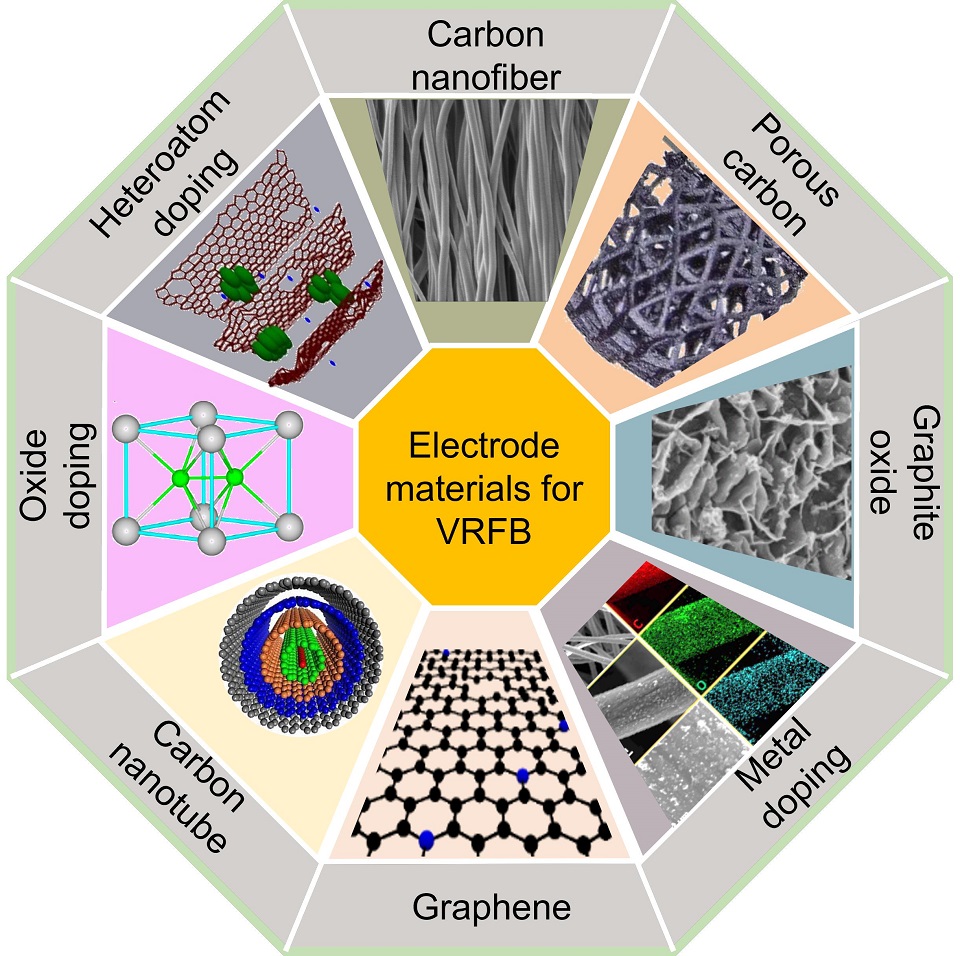
The redox active species in all-vanadium redox flow batteries (VRFBs) reside in the electrolyte, while the heterogeneous reactions occur on the electrode surface; the electrode is therefore the decisive platform for dynamic adsorption, electron transfer, and ion conversion, especially for the VO2+/VO2+ and V2+/V3+ couples. One of the major challenges for VRFB is the slow charge transfer in VO2+/VO2+ and V2+/V3+ reactions, mainly caused by poor catalytic performance of electrodes and weak adhesion of catalysts to electrodes. This review focuses on the key challenges and recent advancements in VRFB. It begins with an overview of VRFB, including their history, working principles, applications, and the advantages and limitations associated with their use. One persistent, under-addressed trade-off is that strategies that boost apparent activity (e.g., high defect density or surface area) can degrade adhesion and cycling durability under flow shear; activity should therefore be co-reported with adhesion and durability descriptors. Addressing this trade-off is critical to improving overall efficiency and stability in VRFB systems. A comprehensive discussion of various electrode materials is presented, categorized by their properties and preparation methods. Special emphasis is placed on the synthesis and application of carbon-based electrode materials, highlighting their potential in addressing these challenges. Finally, we map materials-level gains to stack- and system-level metrics, and outline strategies, with a focus on bifunctional and in-situ grown catalysts, for achieving high-efficiency, high-stability VRFBs.