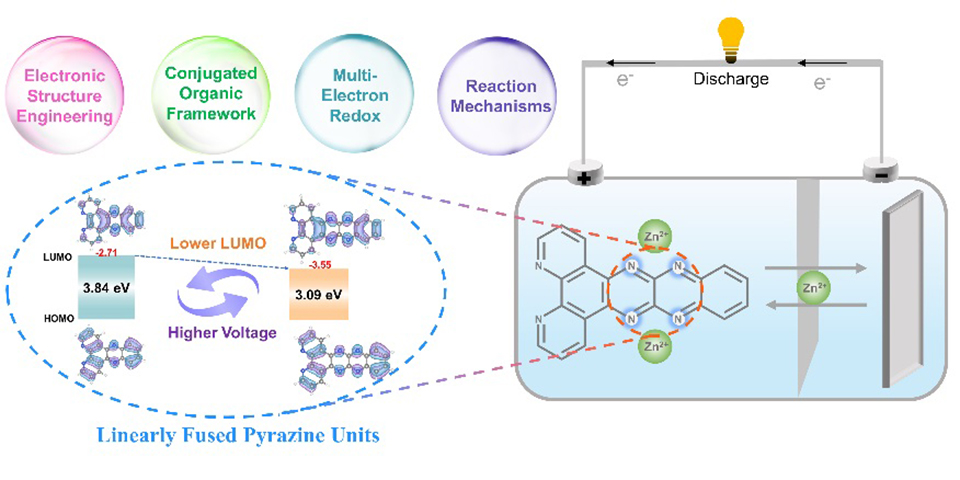
高电压n型有机正极材料是构建高比能、长寿命锌-有机电池(ZOBs)的关键。然而,大多数n型材料的电子结构和较高的电子能级使得n型材料的表现出缓慢的动力学,高溶解性和低放电电压(小于0.8 V)。本文中,我们通过在芳香族化合物的共轭骨架中引入局部缺电子结构,设计了一种小分子,quinoxalino[2',3':5,6]pyrazino[2,3-f][1,10]phenanthroline (DPQP),用于ZOBs正极。由线性稠合的缺电子的吡嗪单元扩展的吡嗪并苯结构优化了材料的电子结构,从而显著提高了材料的放电电压,扩展的共轭平面进一步抑制了活性材料的溶解并促进了快速的电荷转移动力学。得益于这些结果,DPQP电极在0.1A·g-1下的平均工作电压从0.61 V提高到1.07 V(vs. Zn2+/Zn),过电位仅为140 mV,且随着电流密度的增加,未见明显电压降,反应了快速可逆的氧化还原动力学。此外,DPQP正极表现出令人满意的循环性能,在0.1 A·g-1下保持稳定循环超过2000小时并且在10 A·g-1循环超过10000圈且具有82.5%的高容量保持率。值得注意的是,DPQP电极具有出色的极端温度适应性,能够在-20℃-60℃的宽温域中表现出令人满意的电化学性能。同时,通过一系列物理表征证明了DPQP正极高度的电化学可逆性和锌离子存储机制。
High-voltage n-type organic cathode materials are critical for constructing zinc-organic batteries (ZOBs) with high energy density and long cycle life. However, the intrinsically unfavorable electronic structures and relatively high LUMO energy levels of most n-type materials often lead to sluggish kinetics, high solubility, and suboptimal discharge voltages (<0.8 V). Here, we design a small molecule, quinoxalino[2',3':5,6]pyrazino[2,3-f][1,10]phenanthroline (DPQP), as a ZOB cathode by introducing locally electron-deficient motifs into the conjugated backbone of aromatic compounds. The linearly fused pyrazine units extending the pyrazine–benzene framework effectively optimize the electronic structure, thereby significantly enhancing the discharge voltage. Meanwhile, the expanded π-conjugated plane suppresses dissolution and accelerates charge-transfer kinetics. Benefiting from these features, the DPQP electrode exhibits an exceptional increase in average operating voltage from 0.61 V to 1.07 V (vs. Zn2+/Zn) at 0.1 A·g-1, with an overpotential of only 140 mV. Notably, no discernible voltage decay occurs as the current density increases, indicating rapid and highly reversible redox kinetics. Furthermore, the DPQP cathode delivers outstanding cycling stability, maintaining over 2000 h of continuous operation at 0.1 A·g-1 and retaining 82.5% of its capacity after more than 10,000 cycles at 10 A·g-1. Remarkably, the DPQP electrode also demonstrates excellent tolerance to extreme temperatures, achieving stable electrochemical performance across a wide temperature range from -20 °C to 60 °C. In addition, a series of spectroscopic and microscopic characterizations confirm the highly reversible redox behavior and Zn2+ storage mechanism of the DPQP cathode.