

金属/溶液界面的熵:热力学框架、理论模型和界面水的作用
收稿日期: 2025-11-13
修回日期: 2025-12-10
录用日期: 2025-12-24
网络出版日期: 2025-12-24
Entropy of Metal/Solution Interfaces: Thermodynamic Framework, Theoretical Models, and Roles of Interfacial Water
#Equal contributions
Received date: 2025-11-13
Revised date: 2025-12-10
Accepted date: 2025-12-24
Online published: 2025-12-24
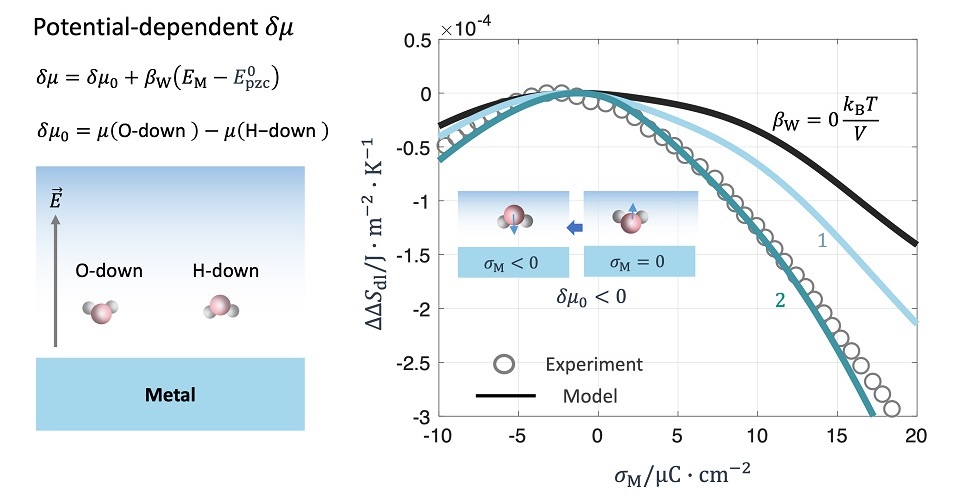
熵是金属/溶液界面双电层的基本热力学性质之一,但其定义、测量方法和理论处理在现有文献中较为分散,且在某些情况下仍存在歧义。本文重新审视了双电层的热力学理论,明确区分并比较了两类熵:过剩熵和形成熵。针对形成熵,在最基本的双电层模型-Gouy-Chapman(GC)模型框架下,验证了两种等价的计算途径。在澄清相关概念和计算方法之后,本文采用改进的Gouy-Chapman-Stern(GCS)模型研究了界面水对双电层形成熵的影响。该模型显式引入了氧端朝向与氢端朝向水分子之间的化学势差,记为δμ。模型计算得到的微分电容和熵与Au(111)电极在水溶液中形成的双电层实验数据进行了对比。结果表明,当未带电表面上水分子更倾向于氧端朝向构型时,最大熵对应的表面荷电(CME)为负值;此外,当δμ随电位变化时,形成熵在CME附近呈现出明显的非对称性。然而,该模型无法同时再现来自两项独立实验研究的电容和熵测量结果,表明模型仍存在不足,或实验数据中可能存在误差。尽管如此,本工作强调了在研究双电层时同步测量电容和熵的重要性。
张增明 , 梁子西 , 黄俊 . 金属/溶液界面的熵:热力学框架、理论模型和界面水的作用[J]. 电化学, 2026 , 32(3) : 2515010 . DOI: 10.61558/2993-074X.3601
Entropy is a basic thermodynamic property of the electrical double layer (EDL) at metal/solution interfaces, yet, its definition, measurement, and theoretical treatment are dispersed in the literature, and, in some cases, ambiguous. In this paper, we revisit the thermodynamic theory of EDL, from which two variants of entropy, excess entropy and formation entropy, are obtained and compared. In terms of the formation entropy, two calculation routes are validated in the context of a primitive EDL model, namely, the Gouy-Chapman (GC) model. After clarifying the concepts and calculation routes, we investigate interfacial water effects on the EDL entropy, using a refined Gouy-Chapman-Stern (GCS) model accounting for chemical potential difference between oxygen- and hydrogen-down water molecules, denoted

| [1] | Helmholtz H. Studien Über Electrische Grenzschichten[J]. Ann. Phys., 1879, 243(7): 337-382. https://doi.org/10.1002/andp.18792430702. |
| [2] | Stern O H. Zur Theorie Der Elektrolytischen Doppelschicht. Z. Für Elektrochem[J]. Angew. Phys. Chem., 1924, 30(21-22): 508-516. https://doi.org/10.1002/bbpc.192400182. |
| [3] | Guidelli R, Aloisi G, Leiva E, Schmickler W. Contribution to the entropy of formation of metal/solution interphases from metal electrons[J]. J. Phys. Chem., 1988, 92(23): 6671-6675. https://doi.org/10.1021/j100334a036. |
| [4] | Conway B E. Electrochemical supercapacitors[M]. Springer US: Boston, MA, 1999. https://doi.org/10.1007/978-1-4757-3058-6. |
| [5] | Schmickler W, Santos E. Interfacial electrochemistry[M]. Springer Berlin Heidelberg: Berlin, Heidelberg, 2010. https://doi.org/10.1007/978-3-642-04937-8. |
| [6] | Bazant M Z, Kilic M S, Storey B D, Ajdari A. Towards an understanding of induced-charge electrokinetics at large applied voltages in concentrated solutions[J]. Adv. Colloid Interface Sci., 2009, 152(1-2): 48-88. https://doi.org/10.1016/j.cis.2009.10.001. |
| [7] | Zeng L, Chen M, Wang Z X, Qiao R, Feng G. Structural evolution governs reversible heat generation in electrical double layers[J]. Phys. Rev. Lett., 2023, 131: 096201. https://doi.org/10.1103/PhysRevLett.131.096201. |
| [8] | Garrick T R, Koch B J, Choi M, Du X N, Adeyinka A M, Staser J A, Choe S Y. Quantifying the entropy and enthalpy of insertion materials for battery applications via the multi-species, multi-reaction model[J]. J. Electrochem. Soc., 2024, 171(2): 23502. https://doi.org/10.1149/1945-7111/ad1d27. |
| [9] | Trasatti S. Structure of the metal/electrolyte solution interface: New data for theory[J]. Electrochim. Acta, 1991, 36(11-12): 1659-1667. https://doi.org/10.1016/0013-4686(91)85023-Z. |
| [10] | Climent V, Coles B A, Compton R G. Laser-induced potential transients on a Au(111) single-crystal electrode. Determination of the potential of maximum entropy of double-layer formation[J]. J. Phys. Chem. B, 2002, 106(20): 5258-5265. https://doi.org/10.1021/jp020054q. |
| [11] | Hamelin A, Stoicoviciu L, Silva F. The temperature dependence of the doublelayer properties of gold faces in perchloric acid solutions: Part I. The (210) Gold Face[J]. J. Electroanal. Chem. Interfacial Electrochem., 1987 229(1-2): 107-214. https://doi.org/10.1016/0022-0728(87)85134-3. |
| [12] | Hamelin A, Stoicoviciu L, Silva F. The temperature dependence of the double-layer properties of gold faces in perchloric acid solutions part II. The (110) gold face[J]. J. Electroanal. Chem. Interfacial Electrochem. 1987, 234: 93-105. https://doi.org/10.1016/0022-0728(87)87043-8. |
| [13] | Hamelin A, Doubova L, Stoicoviciu L, Trasatti S. The temperature dependence of the double layer parameters of the (111) face of silver[J]. J. Electroanal. Chem. Interfacial Electrochem., 1988, 244(1-2): 133-145. https://doi.org/10.1016/0022-0728(88)80101-3. |
| [14] | Garcia-Araez N, Climent V, Feliu J. Separation of temperature effects on double-layer and charge-transfer processes for platinum|solution interphases. entropy of formation of the double layer and absolute molar entropy of adsorbed hydrogen and OH on Pt(111)[J]. J. Phys. Chem. C, 2009, 113(46): 19913-19925. https://doi.org/10.1021/jp904638d. |
| [15] | Randles J E B, Whiteley K S. The temperature dependence of the electrocapillary maximum of mercury[J]. Trans. Faraday Soc., 1956, 52: 1509. https://doi.org/10.1039/tf9565201509. |
| [16] | Hills G J, Payne R. Temperature and pressure dependence of the double layer capacity at the mercury-solution interface[J]. Trans. Faraday Soc., 1965, 61: 326. https://doi.org/10.1039/tf9656100326. |
| [17] | Harrison J A, Randles J E B, Schiffrin D J. The entropy of formation of the mercury-aqueous solution interface and the structure of the inner layer[J]. J. Electroanal. Chem. Interfacial Electrochem., 1973, 48(3): 359-381. https://doi.org/10.1016/S0022-0728(73)80369-9. |
| [18] | Hills G J. Compact double layer as a function of temperature and pressure[J]. J. Phys. Chem., 1969, 73(11): 3591-3597. https://doi.org/10.1021/j100845a010. |
| [19] | Fawcett W R. Liquids, solutions, and interfaces: From classical macroscopic descriptions to modern microscopic details[M]. Oxford University Press, 2004. |
| [20] | Ganassin A, Sebastián P, Climent V, Schuhmann W, Bandarenka A S, Feliu J. On the pH dependence of the potential of maximum entropy of Ir(111) electrodes[J]. Sci. Rep., 2017, 7(1): 1246. https://doi.org/10.1038/s41598-017-01295-1. |
| [21] | Sundararaman R, Schwarz K. Solvent effects determine the sign of the charges of maximum entropy and capacitance at silver electrodes[J]. J. Chem. Phys., 2023, 158(12): 121102. https://doi.org/10.1063/5.0143307. |
| [22] | Doubova, Trasatti. The temperature coefficient of the potential of zero charge of Ag single crystal face electrodes in aqueous electrolyte solution[J]. Condens. Matter Phys., 2001, 4(4): 759. https://doi.org/10.5488/CMP.4.4.759. |
| [23] | Schnur S, Groß A. Properties of metal-water interfaces studied from first principles[J]. New J. Phys., 2009, 11(12): 125003. https://doi.org/10.1088/1367-2630/11/12/125003. |
| [24] | Huang J. Zooming into the inner Helmholtz plane of Pt(111)-aqueous solution interfaces: Chemisorbed water and partially charged ions[J]. JACS Au, 2023, 3(2): 550-564. https://doi.org/10.1021/jacsau.2c00650. |
| [25] | Zhen E F, Chen Y X, Huang J. double-layer capacitance peaks: Origins, ion dependence, and temperature effects[J]. J. Chem. Phys., 2025, 162(14): 144702. https://doi.org/10.1063/5.0251548. |
| [26] | Schmickler W, Guidelli R. The partial charge transfer[J]. Electrochim. Acta, 2014, 127: 489-505. https://doi.org/10.1016/j.electacta.2014.02.057. |
| [27] | Kornyshev A A. Double-layer in ionic liquids: Paradigm change?[J]. J. Phys. Chem. B, 2007, 111(20): 5545-5557. https://doi.org/10.1021/jp067857o. |
| [28] | Zhang M K, Cai J, Chen Y X. On the Electrode charge at the metal/solution interface with specific adsorption[J]. Curr. Opin. Electrochem., 2022, 36: 101161. https://doi.org/10.1016/j.coelec.2022.101161. |
| [29] | Li C Y, Le J B, Wang Y H, Chen S, Yang Z L, Li J F, Cheng J, Tian Z Q. In situ probing electrified interfacial water structures at atomically flat surfaces[J]. Nat. Mater., 2019, 18(7): 697-701. https://doi.org/10.1038/s41563-019-0356-x. |
| [30] | Xu M L, Liu S H, Vijay S, Bligaard T, Kastlunger G. Benchmarking water adsorption on metal surfaces with Ab Initio molecular dynamics[J]. J. Chem. Phys., 2024, 160(24): 244707. https://doi.org/10.1063/5.0205552. |
| [31] | Parsons R. The structure of the mercury-electrolyte interphase in the presence of thiourea[J]. Proc. A, 1961, 261(1304): 79-90. https://doi.org/10.1098/rspa.1961.0062. |
| [32] | Bockris J O, Devanathan M A V, Müller K. On the structure of charged interfaces[J]. Electrochemistry, 1965, 832-863. https://doi.org/10.1016/B978-1-4831-9831-6.50068-0. |
| [33] | Domínguez-Flores F, Kiljunen T, Groß A, Sakong S, Melander M M. Metal-water interface formation: Thermodynamics from Ab Initio molecular dynamics simulations[J]. J. Chem. Phys., 2024, 161(4): 44705. https://doi.org/10.1063/5.0220576. |
| [34] | Gregory J K, Clary D C, Liu K, Brown M G, Saykally R J. The water dipole moment in water clusters[J]. Science, 1997, 275(5301): 814-817. https://doi.org/10.1126/science.275.5301.814. |
| [35] | Adnan A, Behjati S, Félez-Guerrero N, Ojha K, Koper M T M. Tracking the surface structure and the influence of cations and anions on the double-layer region of a Au(111) electrode[J]. Phys. Chem. Chem. Phys., 2024, 26(32): 21419-21428. https://doi.org/10.1039/D4CP02133A. |
| [36] | Lee J, Yeo H, Lee R G, Kim Y H. Ab Initio theory of the nonequilibrium adsorption energy[J]. Npj Comput. Mater., 2024, 10(1): 60. https://doi.org/10.1038/s41524-024-01242-5. |
| [37] | Macdonald J R. Theory of the differential capacitance of the double layer in unadsorbed electrolytes[J]. J. Chem. Phys., 1954, 22(11): 1857-1866. https://doi.org/10.1063/1.1739933. |
| [38] | Braunwarth L, Jung C, Jacob T. Potential‐dependent Pt(111)/water interface: Tackling the challenge of a consistent treatment of electrochemical interfaces[J]. ChemPhysChem, 2023, 24(1): e202200336. https://doi.org/10.1002/cphc.202200336. |
| [39] | Zhang L L, Li C K, Huang J. A beginners’ guide to modelling of electric double layer under equilibrium, nonequilibrium and AC conditions[J]. J Electrochem., 2022, 28(2): 2108471. https://electrochem.xmu.edu.cn/CN/10.13208/j.electrochem.210847. |
/
| 〈 |
|
〉 |