

碳载铂镍八面体纳米粒子作为质子交换膜燃料电池阴极催化剂的性能研究
收稿日期: 2025-07-13
修回日期: 2025-09-07
录用日期: 2025-10-13
网络出版日期: 2025-10-13
Carbon Supported Octahedral PtNi Nanoparticles (Oct-PtNi/C) as a Cathode Catalyst for Proton Exchange Membrane Fuel Cells (PEMFCs) with Improved Activity and Durability
Received date: 2025-07-13
Revised date: 2025-09-07
Accepted date: 2025-10-13
Online published: 2025-10-13
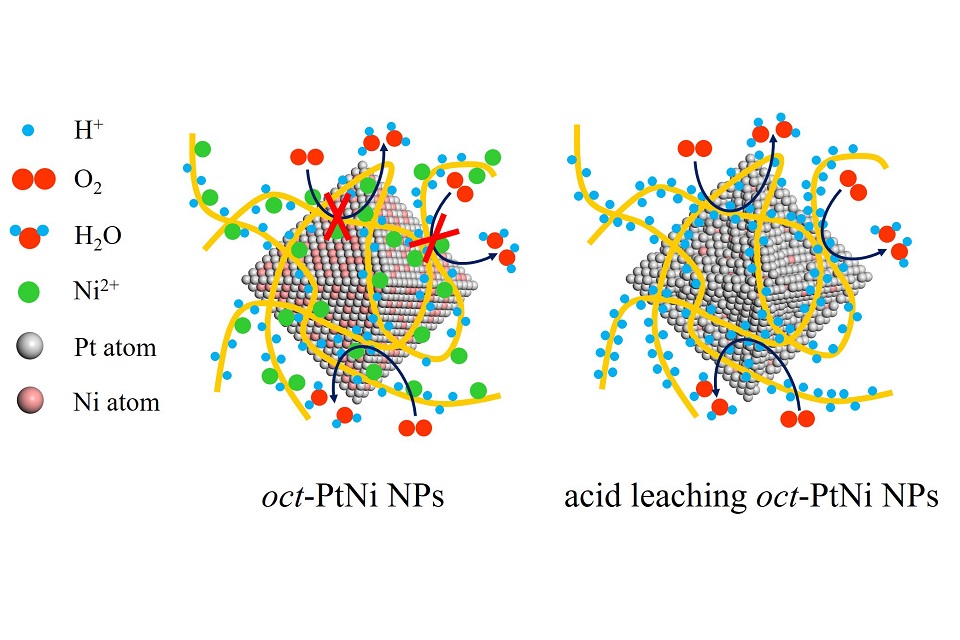
质子交换膜燃料电池(PEMFC)是一种有前景的能量转换装置,但其大规模商业应用受制于高昂的成本和较低的性能。PEMFC的性能主要受限于阴极氧还原反应(ORR)的动力学。铂是对氧还原反应催化活性最高的单金属,但是其性能仍难以满足商业化应用。前期研究表明,铂镍八面体纳米粒子(oct-PtNi-NPs)在半电池中具有优异的ORR活性,但其在膜电极组件(MEA)中的性能报道较少。本论文研究了碳载铂镍八面体纳米粒子(oct-PtNi/C)作为阴极催化剂的膜电极组件性能。研究结果表明,温和的酸洗条件可溶解oct-PtNi/C表面不稳定的镍原子,得到PNC-A催化剂;以PNC-A催化剂为阴极催化剂的MEA性能得到提高:当阴极铂负载量为0.2 mg∙cm-2时,MEA的最大功率密度达到1.0 W∙cm-2,比使用商用Pt/C作为催化剂时高15%;经30,000圈加速老化实验后,使用PNC-A催化剂的MEA的性能保持率为82%,高于Pt/C(74%)。本论文研究了使用PNC-A催化剂作为PEMFC阴极催化剂的可能性,并发现该催化剂提高了PEMFC的性能,同时降低了贵金属铂的用量。
丰紫薇 , 陈海忠 , 段骁 , 唐玲 , 赵云昆 , 黄龙 . 碳载铂镍八面体纳米粒子作为质子交换膜燃料电池阴极催化剂的性能研究[J]. 电化学, 2026 , 32(1) : 2515009 . DOI: 10.61558/2993-074X.3588
Proton exchange membrane fuel cells (PEMFCs) are considered as a promising renewable power source. However, the massive commercial application of PEMFCs has been greatly hindered by their high expense and less-satisfied performance mainly due to the sluggish oxygen reduction reaction (ORR) kinetics even on state-of-the-art Pt catalyst. Octahedral PtNi nanoparticles (oct-PtNi NPs) with excellent ORR activity in a half-cell have been widely studied, while their performance in membrane electrode assembly (MEA) has much less reported. Herein, we investigated the MEA performance using the carbon supported oct-PtNi NPs (oct-PtNi/C) as the cathode catalyst. Under the mild acid washing condition, the surface Ni atoms of oct-PtNi/C were largely removed, and the performance of the MEA using the acid-leaching oct-PtNi/C (PNC-A) as the cathode catalyst was greatly improved. The maximum power density of the MEA reached 1.0 W·cm-2 with the cathode Pt loading of 0.2 mg·cm-2, which is 15% higher than that using Pt/C as the catalyst. After 30k cycles in the accelerated degradation test (ADT), the MEA using PNC-A as the catalyst showed a performance retention of 82%, higher than that of Pt/C (74%). The results reported here verify the possibility of using PNC-A as an advanced cathode catalyst in PEMFCs, thus enhancing the performance of PEMFCs while lowering the amount of expensive Pt.

| [1] | Wang X X, Swihart M T, Wu G. Achievements, challenges and perspectives on cathode catalysts in proton exchange membrane fuel cells for transportation[J]. Nat. Catal., 2019, 2(7): 578-589. http://dx.doi.org/10.1038/s41929-019-0304-9. |
| [2] | Swider Lyons K E, Campbell S A. Physical chemistry research toward proton exchange membrane fuel cell advancement[J]. J. Phys. Chem. Lett., 2013, 4(3): 393-401. http://dx.doi.org/10.1021/jz3019012. |
| [3] | Liu Z, Peng B, Tsai Y H J, Zhang A, Xu M, Zang W, Yan X, Xing L, Pan X, Duan X, Huang Y. Pt catalyst protected by graphene nanopockets enables lifetimes of over 200,000?h for heavy-duty fuel cell applications[J]. Nat. Nanotechnol., 2025, 20: 807-814. http://dx.doi.org/10.1038/s41565-025-01895-3. |
| [4] | Shao M, Peles A, Shoemaker K. Electrocatalysis on platinum nanoparticles: Particle Size effect on oxygen reduction reaction activity[J]. Nano Lett., 2011, 11(9): 3714-3719. http://dx.doi.org/10.1021/nl2017459. |
| [5] | Hernandez Fernandez P, Masini F, McCarthy D N, Strebel C E, Friebel D, Deiana D, Malacrida P, Nierhoff A, Bodin A, Wise A M, Nielsen J H, Hansen T W, Nilsson A, StephensIfan E L, Chorkendorff I. Mass-selected nanoparticles of PtxY as model catalysts for oxygen electroreduction[J]. Nat Chem., 2014, 6(8): 732-738. http://dx.doi.org/10.1038/nchem.2001. |
| [6] | Chen C, Kang Y, Huo Z, Zhu Z, Huang W, Xin H L, Snyder J D, Li D, Herron J A, Mavrikakis M, Chi M, More K L, Li Y, Markovic N M, Somorjai G A, Yang P, Stamenkovic V R. Highly crystalline multimetallic nanoframes with three-dimensional electrocatalytic surfaces[J]. Science, 2014, 343(6177): 1339-1343. http://dx.doi.org/doi:10.1126/science.1249061. . |
| [7] | Cui C, Gan L, Heggen M, Rudi S, Strasser P. Compositional segregation in shaped Pt alloy nanoparticles and their structural behaviour during electrocatalysis[J]. Nat. Mater., 2013, 12(8): 765-771. http://dx.doi.org/10.1038/nmat3668. |
| [8] | Wang C, Chi M, Li D, Strmcnik D, van der Vliet D, Wang G, Komanicky V, Chang K C, Paulikas A P, Tripkovic D, Pearson J, More K L, Markovic N M, Stamenkovic V R. Design and synthesis of bimetallic electrocatalyst with multilayered Pt-skin surfaces[J]. J. Am. Chem. Soc., 2011, 133(36): 14396-14403. http://dx.doi.org/10.1021/ja2047655. |
| [9] | Polani S, Amitrano R, Baumunk A F, Pan L, Lu J, Schmitt N, Gernert U, Klingenhof M, Selve S, Günther C M, Etzold B J M, Strasser P. Oxygen reduction reaction activity and stability of shaped metal-doped PtNi electrocatalysts evaluated in gas diffusion electrode half-cells[J]. ACS Appl. Mater. Interfaces., 2024, 16(39): 52406-52413. http://dx.doi.org/10.1021/acsami.4c11068. |
| [10] | Fikry M, Wei? N, Bozzetti M, ünsal S, Georgi M, Khavlyuk P, Herranz J, Tileli V, Eychmüller A, Schmidt T J. Up-scaled preparation of Pt-Ni aerogel catalyst layers for polymer electrolyte fuel cell cathodes[J]. ACS Appl. Energy Mater., 2024, 7: 896-905. http://dx.doi.org/10.1021/acsaem.3c01930. |
| [11] | Polani S, MacArthur K E, Klingenhof M, Wang X, Paciok P, Pan L, Feng Q, Kormányos A, Cherevko S, Heggen M, Strasser P. Size and composition dependence of oxygen reduction reaction catalytic activities of Mo-doped PtNi/C octahedral nanocrystals[J]. ACS Catal., 2021, 11(18): 11407-11415. http://dx.doi.org/10.1021/acscatal.1c01761. |
| [12] | Lu B A, Shen L F, Liu J, Zhang Q, Wan L Y, Morris D J, Wang R X, Zhou Z Y, Li G, Sheng T, Gu L, Zhang P, Tian N, Sun S G. Structurally disordered phosphorus-doped Pt as a highly active electrocatalyst for an oxygen reduction reaction[J]. ACS Catal., 2021, 11(1): 355-363. http://dx.doi.org/10.1021/acscatal.0c03137. |
| [13] | Zhao X, Xi C, Zhang R, Song L, Wang C, Spendelow J S, Frenkel A I, Yang J, Xin H L, Sasaki K. High-performance nitrogen-doped intermetallic PtNi catalyst for the oxygen Reduction Reaction[J]. ACS Catal., 2020, 10(18): 10637-10645. http://dx.doi.org/10.1021/acscatal.0c03036. |
| [14] | Yang T, Cheng C, Xiao L, Wang M, Zhang F, Wang J, Yin P, Shen G, Yang J, Dong C, Liu H, Du X. A descriptor of IB alloy catalysts for hydrogen evolution reaction[J]. SmartMat, 2024, 5(3): e1204. http://dx.doi.org/https://doi.org/10.1002/smm2.1204. |
| [15] | Chen J, Arce Ramos J M, Katsounaros I, de Smit E, Abubakar S M, Lum Y, Zhang J, Wang L. Modulating oxygen affinity to enhance liquid products for the electrochemical reduction of carbon monoxide[J]. 2025, 6(2): e70010. http://dx.doi.org/https://doi.org/10.1002/smm2.70010. |
| [16] | Wang C C, Guo Z S, Shen Q, Xu Y R, Lin C P, Yang X D, Li C C, Sun Y Q, Hang L F. Recent advances in core-shell structured noble metal-based catalysts for electrocatalysis[J]. Rare Metals, 2025, 44(4): 2180-2207. http://dx.doi.org/10.1007/s12598-024-03081-1. |
| [17] | Huang X Q, Zhao Z P, Cao L, Chen Y, Zhu E B, Lin Z Y, Li M F, Yan A, Zettl A, Wang Y M, Duan X F, Mueller T, Huang Y. High-performance transition metal-doped Pt3Ni octahedra for oxygen reduction reaction[J]. Science, 2015, 348(6240): 1230-1234. http://dx.doi.org/doi:10.1126/science.aaa8765. |
| [18] | Cui C, Gan L, Li H H, Yu S H, Heggen M, Strasser P. Octahedral PtNi nanoparticle catalysts: exceptional oxygen reduction activity by tuning the alloy particle surface composition[J]. Nano Lett., 2012, 12(11): 5885-5889. http://dx.doi.org/10.1021/nl3032795. |
| [19] | Stamenkovic V R, Fowler B, Mun B S, Wang G, Ross P N, Lucas C A, Markovi? N M. Improved oxygen reduction activity on Pt3Ni(111) via increased surface site availability[J]. Science, 2007, 315(5811): 493-497. http://dx.doi.org/doi:10.1126/science.1135941. |
| [20] | Dionigi F, Weber C C, Primbs M, Gocyla M, Bonastre A M, Sp?ri C, Schmies H, Hornberger E, Kühl S, Drnec J, Heggen M, Sharman J, Dunin-Borkowski R E, Strasser P. Controlling near-surface Ni composition in octahedral PtNi(Mo) nanoparticles by Mo doping for a highly active oxygen reduction reaction catalyst[J]. Nano Lett., 2019, 19(10): 6876-6885. http://dx.doi.org/10.1021/acs.nanolett.9b02116. |
| [21] | Kim O H, Ahn C Y, Kang S Y, Kim S, Choi H J, Cho Y H, Sung Y E. From half-cells to membrane-electrode assemblies: a comparison of oxygen reduction reaction catalyst Performance Characteristics[J]. Fuel Cells, 2019, 19(6): 695-707. http://dx.doi.org/https://doi.org/10.1002/fuce.201900120. |
| [22] | Jia Q, Li J, Caldwell K, Ramaker D E, Ziegelbauer J M, Kukreja R S, Kongkanand A, Mukerjee S. Circumventing metal dissolution induced degradation of Pt-alloy catalysts in proton exchange membrane fuel cells: revealing the asymmetric volcano nature of redox catalysis[J]. ACS Catal., 2016, 6(2): 928-938. http://dx.doi.org/10.1021/acscatal.5b02750. |
| [23] | Han B, Carlton C E, Kongkanand A, Kukreja R S, Theobald B R, Gan L, O'Malley R, Strasser P, Wagner F T, Shao-Horn Y. Record activity and stability of dealloyed bimetallic catalysts for proton exchange membrane fuel cells[J]. Energy Environ. Sci., 2015, 8(1): 258-266. http://dx.doi.org/10.1039/C4EE02144D. |
| [24] | Wang J, Xue Q, Li B, Yang D, Lv H, Xiao Q, Ming P, Wei X, Zhang C. Preparation of a graphitized-carbon-supported PtNi octahedral catalyst and application in a proton-exchange membrane fuel cell[J]. ACS Appl. Mater. Interfaces., 2020, 12(6): 7047-7056. http://dx.doi.org/10.1021/acsami.9b17248. |
| [25] | Wang J, Li B, Gao X, Yang D J, Lv H, Xiao Q F, Zhang C M. From rotating disk electrode to single cell: Exploration of PtNi/C octahedral nanocrystal as practical proton exchange membrane fuel cell cathode catalyst[J]. J. Power. Sources, 2018, 406: 118-127. http://dx.doi.org/https://doi.org/10.1016/j.jpowsour.2018.10.010. |
| [26] | Huang X Q, Zhao Z P, Chen Y, Zhu E B, Li M F, Duan X F, Huang Y. A rational design of carbon-supported dispersive Pt-based octahedra as efficient oxygen reduction reaction catalysts[J]. Energy Environ. Sci., 2014, 7(9): 2957-2962. http://dx.doi.org/10.1039/C4EE01082E. |
| [27] | FCTT AST and polarization curve protocols for PEMFCs[EB/OL]. https://uscar.org/technologies-teams/hydrogen-fuel-cell. |
| [28] | Gasteiger H A, Kocha S S, Sompalli B, Wagner F T. Activity benchmarks and requirements for Pt, Pt-alloy, and non-Pt oxygen reduction catalysts for PEMFCs[J]. App. Catal. B: Environ., 2005, 56(1-2): 9-35. http://dx.doi.org/https://doi.org/10.1016/j.apcatb.2004.06.021. |
| [29] | Tang M H, Zhang S M, Chen S L. Pt utilization in proton exchange membrane fuel cells: structure impacting factors and mechanistic insights[J]. Chem. Soc. Rev., 2022, 51(4): 1529-1546. http://dx.doi.org/10.1039/D1CS00981H. |
| [30] | Gan L, Heggen M, O’Malley R, Theobald B, Strasser P. Understanding and controlling nanoporosity formation for improving the stability of bimetallic fuel cell catalysts[J]. Nano Lett., 2013, 13(3): 1131-1138. http://dx.doi.org/10.1021/nl304488q. |
| [31] | Vliet D F, Wang C, Li D, Paulikas A P, Greeley J, Rankin R B, Strmcnik D, Tripkovic D, Markovic N M, Stamenkovic V R. Unique electrochemical adsorption properties of Pt-skin surfaces[J]. Angew. Chem. Int. Ed., 2012, 51(13): 3139-3142. http://dx.doi.org/https://doi.org/10.1002/anie.201107668. |
| [32] | Khalakhan I, Bogar M, Vorokhta M, Kú? P, Yakovlev Y, Dopita M, Sandbeck D J S, Cherevko S, Matolínová I, Amenitsch H. Evolution of the PtNi bimetallic alloy fuel cell catalyst under simulated operational conditions[J]. ACS Appl. Mater. Interfaces,. 2020, 12(15): 17602-17610. http://dx.doi.org/10.1021/acsami.0c02083. |
| [33] | Kelly M J, Egger B, Fafilek G, Besenhard J O, Kronberger H, Nauer G E. Conductivity of polymer electrolyte membranes by impedance spectroscopy with microelectrodes[J]. Solid State Ion., 2005, 176(25): 2111-2114. http://dx.doi.org/https://doi.org/10.1016/j.ssi.2004.07.071. |
| [34] | Makharia R, Mathias M F, Baker D R. Measurement of catalyst layer electrolyte Resistance in PEFCs using electrochemical impedance spectroscopy[J]. J. Electrochem. Soc., 2005, 152(5): A970-A977. http://dx.doi.org/10.1149/1.1888367. |
| [35] | Sharma R, Gyergyek S, Li Q, Andersen S M. Evolution of the degradation mechanisms with the number of stress cycles during an accelerated stress test of carbon supported platinum nanoparticles[J]. J. Electroanal. Chem., 2019, 838: 82-88. http://dx.doi.org/https://doi.org/10.1016/j.jelechem.2019.02.052. |
| [36] | Greszler T A, Caulk D, Sinha P. The impact of platinum loading on oxygen transport resistance[J]. J. Electrochem. Soc., 2012, 159(12): F831-F840. http://dx.doi.org/10.1149/2.061212jes. |
/
| 〈 |
|
〉 |