

水系钠离子电池钛酸亚磷酸盐阳极:进展和展望
收稿日期: 2025-06-08
修回日期: 2025-07-26
录用日期: 2025-08-19
网络出版日期: 2025-08-19
The NTP Anode for Aqueous Sodium Ion Batteries: Recent Advances and Future Perspectives
Received date: 2025-06-08
Revised date: 2025-07-26
Accepted date: 2025-08-19
Online published: 2025-08-19
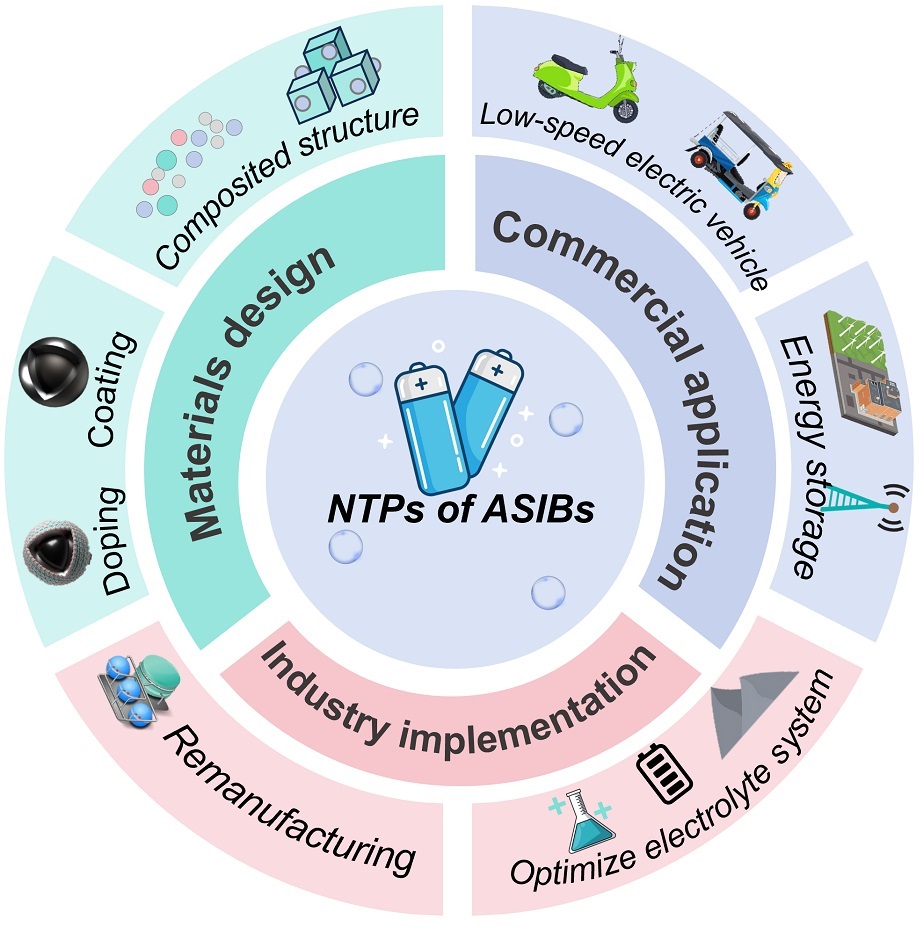
水系钠离子电池因其较高的安全性而在水系电池领域备受关注。然而,水基电解液的特性降低了负极材料工作电位以及电化学稳定性,进而阻碍了水系钠离子电池的大规模应用。钛酸亚磷酸盐(NaTi2(PO4)3,NTP)因其出色的电化学性能和可调的结构,被认为是最具实用化前景的用于水系钠离子电池的负极极材料之一。近年来,围绕NTP的研究取得了显著进展,但关于其研究现状和未来发展方向的综述仍然缺乏。在此背景下,本文首先介绍了NTP的基本特性,并深入分析了其实际应用所面临的挑战。随后,全面概述了提升NTP电化学性能的改性策略。最后,基于当前的研究状况和实际需求,提出了推动实现水系钠离子电池实际应用的建议和展望。本综述旨在为未来研究提供方向指引,推动从基础材料创新逐步过渡到工业应用,进而加速水系钠离子电池的大规模商业化进程。
王明理 , 苏雪颖 , 单政翔 , 杨书哲 , 郭恒瑞 , 罗浩 , 晁栋梁 . 水系钠离子电池钛酸亚磷酸盐阳极:进展和展望[J]. 电化学, 2026 , 32(1) : 2515008 . DOI: 10.61558/2993-074X.3579
Aqueous sodium-ion batteries (ASIBs) have attracted great attention in aqueous batteries due to their merit of high safety. However, the constrained work potential and insufficient chemical stability of anode materials in aqueous electrolytes hinder the large-scale application of ASIBs. Sodium titanium phosphate, NaTi2(PO4)3 (NTP), is considered one of the most promising anode materials for ASIBs due to its excellent electrochemical performance and tunable structure. Recently, great achievements have been made in the development of NTP, however, a comprehensive review of existing studies is still lacking. This article firstly introduces the basic properties of NTP and analyzes the existing challenges. Subsequently, it will provide a comprehensive overview of the key strategies related to the design and modification of NTP materials with optimized electrochemical performance. Finally, based on the current research status and practical needs, suggestions, and future perspectives for advancing NTP in practical applications of ASIBs are presented. This review aims to guide the future research trajectory from basic material innovation to industrial applications, thus promoting the large-scale commercialization of ASIBs.

Key words: Aqueous sodium ion battery; Anode material; NaTi2(PO4)3; Energy storage
| [1] | Tao Z, Chen J. Secondary battery systems for energy storage in smart grids[J]. Chin. Sci. Bull., 2012, 57(27): 2545-2560. https://doi.org/10.1360/972011-2190. |
| [2] | Dunn B, Kamath H, Tarascon J M. Electrical energy storage for the grid: A battery of choices[J]. Science, 2011, 334(6058): 928-935. https://doi.org/10.1126/science.1212741. |
| [3] | Wang Y G, Yi J, Xia Y Y. Recent progress in aqueous lithium-ion batteries[J]. Adv. Energy Mater., 2012, 2(7): 830-840. https://doi.org/10.1002/aenm.201200065. |
| [4] | Kundu D, Talaie E, Duffort V, Nazar L F. The emerging chemistry of sodium ion batteries for electrochemical energy storage[J]. Angew. Chem. Int. Ed., 2015, 54(11): 3431-3448. https://doi.org/10.1002/anie.201410376. |
| [5] | Yu L, Li J, Wang G R, Peng B, Liu R, Shi L, Zhang G Q. Rational design of unique mose-carbon nanobowl particles endows superior alkali metal-ion storage beyond lithium[J]. ACS Appl. Mater. Interfaces, 2021, 13(51): 61116-61128. https://doi.org/10.1021/acsami.1c18234. |
| [6] | Hu X J, Gao F F, Xiao Y, Wang D P, Gao Z H, Huang Z F, Ren S D, Jiang N, Wu S T. Advancements in the safety of lithium-ion battery: The trigger, consequence and mitigation method of thermal runaway[J]. Chem. Eng. J., 2024, 481: 148450. https://doi.org/10.1016/j.cej.2023.148450. |
| [7] | Xu J J, Cai X Y, Cai S M, Shao Y X, Hu C, Lu S R, Ding S. High-energy lithium-ion batteries: Recent progress and a promising future in applications[J]. Energy Environ. Mater., 2023, 6(5): 12450. https://doi.org/10.1002/eem2.12450. |
| [8] | Bin D, Wang F, Tamirat A G, Suo L M, Wang Y G, Wang C S, Xia Y Y. Progress in aqueous rechargeable sodium-ion batteries[J]. Adv. Energy Mater., 2018, 8(17): 1703008. https://doi.org/10.1002/aenm.201703008. |
| [9] | Zhang H, Liu X, Li H H, Hasa I, Passerini S. Challenges and strategies for high-energy aqueous electrolyte rechargeable batteries[J]. Angew. Chem. Int. Ed., 2021, 60(2): 598-616. https://doi.org/10.1002/anie.202004433. |
| [10] | Yue J M, Suo L M. Progress in rechargeable aqueous alkali-ion batteries in china[J]. Energ. Fuel., 2021, 35(11): 9228-9239. https://doi.org/10.1021/acs.energyfuels.1c00817. |
| [11] | Liu M, Ao H, Jin Y, Hou Z, Zhang X, Zhu Y, Qian Y. Aqueous rechargeable sodium ion batteries: Developments and prospects[J]. Mater. Today Energy, 2020, 17: 100432. https://doi.org/10.1016/j.mtener.2020.100432. |
| [12] | Suo L, Borodin O, Wang Y, Rong X, Sun W, Fan X, Xu S, Schroeder M A, Cresce A V, Wang F, Yang C, Hu Y S, Xu K, Wang C. “Water-in-salt” electrolyte makes aqueous sodium-ion battery safe, green, and long-lasting[J]. Adv. Energy Mater., 2017, 7(21): 1701189. https://doi.org/10.1002/aenm.201701189. |
| [13] | Liu J L, Xu C H, Chen Z, Ni S B, Shen Z X. Progress in aqueous rechargeable batteries[J]. Green Energy Environ., 2018, 3(1): 20-41. https://doi.org/10.1016/j.gee.2017.10.001. |
| [14] | Huang Y X, Zhao L Z, Li L, Xie M, Wu F, Chen R J. Electrolytes and electrolyte/electrode interfaces in sodium-ion batteries: From scientific research to practical application[J]. Adv. Mater., 2019, 31(21): 1808393. https://doi.org/10.1002/adma.201808393. |
| [15] | Pascal G E, Fouletier M. Electrochemical intercalation of sodium in graphite[J]. Solid State Ionics, 1988, 28: 1172-1175. https://doi.org/10.1016/0167-2738(88)90351-7. |
| [16] | Whitacre J F, Tevar A, Sharma S. Na4Mn9O18 as a positive electrode material for an aqueous electrolyte sodium-ion energy storage device[J]. Electrochem. Commun., 2010, 12(3): 463-466. https://doi.org/10.1016/j.elecom.2010.01.020. |
| [17] | Song W X, Ji X B, Zhu Y R, Zhu H J, Li F Q, Chen J, Lu F, Yao Y P, Banks C E. Aqueous sodium-ion battery using a Na3V2(PO4)3 electrode[J]. Chemelectrochem, 2014, 1(5): 871-876. https://doi.org/10.1002/celc.201300248. |
| [18] | Hou Z G, Zhang X Q, Chen J W, Qian Y T, Chen L F, Lee P S. Towards high-performance aqueous sodium ion batteries: Constructing hollow NaTi2(PO4)3@C nanocube anode with Zn metal-induced pre-sodiation and deep eutectic electrolyte[J]. Adv. Energy Mater., 2022, 12(14): 2104053. https://doi.org/10.1002/aenm.202104053. |
| [19] | Zhao Y, He J, Hu L T, Yang J, Yan C, Shi M J. Carboxyl-substituted organic molecule assembled with Mxene nanosheets for boosting aqueous Na+ storage[J]. Small, 2023, 19(47): 2304182. https://doi.org/10.1002/smll.202304182. |
| [20] | Sawicki M, Shaw L L. Advances and challenges of sodium ion batteries as post lithium ion batteries[J]. RSC Adv., 2015, 5(65): 53129-53154. https://doi.org/10.1039/C5RA08321D. |
| [21] | Gong D C, Wei C Y, Liang Z Z, Tang Y B. Recent advances on sodium‐ion batteries and sodium dual‐ion batteries: State‐of‐the‐art Na+ host anode materials[J]. Small Sci., 2021, 1(6): 2100014. https://doi.org/10.1002/smsc.202100014. |
| [22] | Qiao S Y, Zhou Q W, Ma M, Liu H K, Dou S X, Chong S K. Advanced anode materials for rechargeable sodium-ion batteries[J]. ACS Nano, 2023, 17(12): 11220-11252. https://doi.org/10.1021/acsnano.3c02892. |
| [23] | Yang M R, Luo J, Guo X N, Chen J C, Cao Y L, Chen W H. Aqueous rechargeable sodium-ion batteries: From liquid to hydrogel[J]. Batteries-Basel, 2022, 8(10): 8100180. https://doi.org/10.3390/batteries8100180. |
| [24] | Xu C L, Liu Y, Han S, Chen Z, Ma Y Z, Guo Q B, Zhang P, Yang W Q, Yang C, Zhao J M, Hu Y S. Rational design of aqueous Na ion batteries toward high energy density and long cycle life[J]. J. Am. Chem. Soc., 2025, 147(8): 7039-7049. https://doi.org/10.1021/jacs.4c18168. |
| [25] | Qiu Y G, Yu Y Y, Xu J, Liu Y, Ou M Y, Sun S X, Wei P, Deng Z, Xu Y, Fang C, Li Q, Han J T, Huang Y H. Redox potential regulation toward suppressing hydrogen evolution in aqueous sodium-ion batteries: Na1.5Ti1.5Fe0.5(PO4)3[J]. J. Mater. Chem. A., 2019, 7(43): 24953-24963. https://doi.org/10.1039/C9TA08829F. |
| [26] | Yao H, Gao Y, Lin X H, Zhang H, Li L, Chou S L. Prussian blue analogues for aqueous sodium-ion batteries: Progress and commercialization assessment[J]. Adv. Energy Mater., 2024, 14(32): 2401984. https://doi.org/10.1002/aenm.202401984. |
| [27] | Zhang Y, Yuan C L, Ye K, Jiang X, Yin J L, Wang G L, Cao D X. An aqueous capacitor battery hybrid device based on Na-ion insertion-deinsertion in λ-MnO2 positive electrode[J]. Electrochim. Acta, 2014, 148: 237-243. https://doi.org/10.1016/j.electacta.2014.10.052. |
| [28] | Rako?evi? L, ?trbac S, Poto?nik J, Popovi? M, Jugovi? D, Simatovi? I S. The NaxMnO2 materials prepared by a glycine-nitrate method as advanced cathode materials for aqueous sodium-ion rechargeable batteries[J]. Ceram. Int., 2021, 47(4): 4595-4603. https://doi.org/10.1016/j.ceramint.2020.10.025. |
| [29] | Yang W S, Wang B, Chen Q, Zhao Q, Zhang Q, Lu S S, Gao Y, Wang X X, Xie Q, Ruan Y J. Unravelling capacity fading mechanisms in sodium vanadyl phosphate for aqueous sodium-ion batteries[J]. J. Colloid Interface Sci., 2022, 627: 913-921. https://doi.org/10.1016/j.jcis.2022.07.108. |
| [30] | Wang Y Y, Liu D, Sun M L, Liu J P. Recent progress in electrode materials for aqueous sodium and potassium ion batteries[J]. Mater. Chem. Front., 2021, 5(20): 7384-7402. https://doi.org/10.1039/D1QM01011E. |
| [31] | Riza S A, Xu R G, Liu Q, Hassan M, Yang Q, Mu D B, Li L, Wu F, Key R B. A review of anode materials for sodium ion batteries[J]. New Carbon Mater., 2024, 39(5): 743-769. https://doi.org/10.1016/S1872-5805(24)60886-3. |
| [32] | Jia Q X, Li Z Y, Ruan H L, Luo D W, Wang J J, Ding Z Y, Chen L N. A review of carbon anode materials for sodium-ion batteries: Key materials, sodium-storage mechanisms, applications, and large-scale design principles[J]. Molecules, 2024, 29(18): 4331. https://doi.org/10.3390/molecules29184331. |
| [33] | Chang G L, Zhao Y F, Dong L, Wilkinson D P, Zhang L, Shao Q S, Yan W, Sun X L, Zhang J J. A review of phosphorus and phosphides as anode materials for advanced sodium-ion batteries[J]. J. Mater. Chem. A., 2020, 8(10): 4996-5048. https://doi.org/10.1039/C9TA12169B. |
| [34] | Balogun M S, Luo Y, Qiu W T, Liu P, Tong Y X. A review of carbon materials and their composites with alloy metals for sodium ion battery anodes[J]. Carbon, 2016, 98: 162-178. https://doi.org/10.1016/j.carbon.2015.09.091. |
| [35] | He B, Yin K B, Gong W B, Xiong Y W, Zhang Q C, Yang J, Wang Z X, Wang Z, Chen M X, Man P, Coquet P, Yao Y G, Sun L T, Wei L. NaTi2(PO4)3 hollow nanoparticles encapsulated in carbon nanofibers as novel anodes for flexible aqueous rechargeable sodium-ion batteries[J]. Nano Energy, 2021, 82: 105764. https://doi.org/10.1016/j.nanoen.2021.105764. |
| [36] | Rui X, Zhang X, Xu S, Tan H, Jiang Y, Gan L Y, Feng Y, Li C C, Yu Y. A low‐temperature sodium‐ion full battery: Superb kinetics and cycling stability[J]. Adv. Funct. Mater., 2020, 31(11): 1616-3028. https://doi.org/10.1002/adfm.202009458. |
| [37] | Patra B, Hegde R, Natarajan A, Deb D, Sachdeva D, Ravishankar N, Kumar K, Gautam G S, Senguttuvan P. Stabilizing multi-electron NASICON-Na1.5N0.5Nb1.5(PO4)3 anode via structural modulation for long-life sodium-ion batteries[J]. Adv. Energy Mater., 2024, 14(17): 2304091. https://doi.org/10.1002/aenm.202304091. |
| [38] | Xu G B, Yang L Y, Yan Z H, Huang Z K, Li X, Guo G C, Tian Y, Yang L W, Huang J Y, Liang Y R, Chou S L. Multiscale structural NaTi2(PO4)3 anode for sodium-ion batteries with long cycle, high areal capacity, and wide operation temperature[J]. Carbon Energ., 2024, 6(10): 2552. https://doi.org/10.1002/cey2.552. |
| [39] | Pang G, Yuan C Z, Nie P, Ding B, Zhu J J, Zhang X G. Synthesis of NASICON-type structured NaTi2(PO4)3-graphene nanocomposite as an anode for aqueous rechargeable Na-ion batteries[J]. Nanoscale, 2014, 6(12): 6328-6334. https://doi.org/10.1039/C3NR06730K. |
| [40] | Chen S Q, Wu C, Shen L F, Zhu C B, Huang Y Y, Xi K, Maier J, Yu Y. Challenges and perspectives for NASICON-type electrode materials for advanced sodium-ion batteries[J]. Adv. Mater., 2017, 29(48): 1700431. https://doi.org/10.1002/adma.201700431. |
| [41] | Li X N, Zhu X B, Liang J W, Hou Z G, Wang Y, Lin N, Zhu Y C, Qian Y T. Graphene-supported NaTi2(PO4)3 as a high rate anode material for aqueous sodium ion batteries[J]. J. Electrochem. Soc., 2014, 161(6): A1181-A1187. https://doi.org/10.1149/2.0081409jes. |
| [42] | Wu W, Yan J Y, Wise A, Rutt A, Whitacre J F. Using intimate carbon to enhance the performance of NaTi2(PO4)3 anode materials: Carbon nanotubes vs Graphite[J]. J. Electrochem. Soc., 2014, 161(4): A561-A567. https://doi.org/10.1149/2.059404jes. |
| [43] | Mohamed A I, Whitacre J F. Capacity fade of NaTi2(PO4)3 in aqueous electrolyte solutions: Relating pH increases to long term stability[J]. Electrochim. Acta, 2017, 235: 730-739. https://doi.org/10.1016/j.electacta.2017.03.106. |
| [44] | Kim H, Hong J, Park K Y, Kim H, Kim S W, Kang K. Aqueous rechargeable Li and Na ion batteries[J]. Chem. Rev., 2014, 114(23): 11788-11827. https://doi.org/10.1021/cr500232y. |
| [45] | Lei P, Liu K L, Wan X, Luo D X, Xiang X D. Ultrafast Na intercalation chemistry of Na2Ti3/2Mn1/2(PO4)3 nanodots planted in a carbon matrix as a low cost anode for aqueous sodium-ion batteries[J]. Chem. Commun., 2019, 55(4): 509-512. https://doi.org/10.1039/C8CC07668E. |
| [46] | Cao F T, Shan X L, Wu J T, Chen Y X, Zhou Y, Wang W J, Zeng C L. Toward suppressing hydrogen evolution with enhanced performance for Bi‐modified NaTi2(PO4)3 anodes in aqueous Na‐ion batteries[J]. Batteries Supercaps., 2025: e202400767. https://doi.org/10.1002/batt.202400767. |
| [47] | Hung T F, Lan W H, Yeh Y W, Chang W S, Yang C C, Lin J C. Hydrothermal synthesis of sodium titanium phosphate nanoparticles as efficient anode materials for aqueous sodium-ion batteries[J]. ACS Sustain. Chem. Eng., 2016, 4(12): 7074-7079. https://doi.org/10.1021/acssuschemeng.6b01962. |
| [48] | Zhang F, Li W F, Xiang X D, Sun M L. Nanocrystal‐assembled porous Na3MgTi(PO4)3 aggregates as highly stable anode for aqueous sodium‐ion batteries[J]. Chem-Eur J., 2017, 23(52): 12944-12948. https://doi.org/10.1002/chem.201703044. |
| [49] | Xu T, Zhao M S, Li Z, Su Z, Ren W, Yang S, Pol V G. A high rate and long cycling performance NaTi2(PO4)3 core-shell porous nanosphere anode for aqueous sodium-ion batteries[J]. Energy Technol., 2022, 10(11): 2200970. https://doi.org/10.1002/ente.202200970. |
| [50] | Ding B, Li M Z, Zheng F Z, Ma Y Z, Song G S, Guan X L, Cao Y, Wen C. Synthesis and performance of NaTi2(PO4)3/VGCF@C anode composite material for aqueous sodium-ion batteries[J]. Batteries, 2023, 9(5): 265. https://doi.org/10.3390/batteries9050265. |
| [51] | Lu M L, Li T, Yang X Q, Liu Y, Xiang X D. A liquid-phase reaction strategy to construct aqueous sodium-ion batteries anode with enhanced redox reversibility and cycling stability[J]. Chem. Eng. Sci., 2023, 274: 118700. https://doi.org/10.1016/j.ces.2023.118700. |
| [52] | Liu Z X, An Y F, Pang G, Dong S Y, Xu C Y, Mi C H, Zhang X G. Tin modified NaTi2(PO4)3as an anode material for aqueous sodium ion batteries[J]. Chem. Eng. J., 2018, 353: 814-823. https://doi.org/10.1016/j.cej.2018.07.159. |
| [53] | Stüwe T, Werner D, Stock D, Thurner C W, Th?ny A, Grie?er C, Loerting T, Portenkirchner E. Enhanced electrochemical performance of NTP/C with rutile TiO2 coating, as anode material for sodium‐ion batteries[J]. Batteries Supercaps., 2023, 6(10): e202300228. https://doi.org/10.1002/batt.202300228. |
| [54] | Kitajou A, Yamashita M, Kobayashi W, Okada M, Nanami T, Muto S. Anode properties of NaTi2(PO4)3 prepared by adding excess Na2CO3 for aqueous sodium-ion batteries[J]. ACS Appl. Energy Mater., 2022, 5(8): 9587-9594. https://doi.org/10.1021/acsaem.2c01212. |
| [55] | He B, Man P, Zhang Q C, Fu H L, Zhou Z Y, Li C W, Li Q L, Wei L, Yao Y G. All binder-free electrodes for high-performance wearable aqueous rechargeable sodium-ion batteries[J]. Nano-Micro Lett., 2019, 11(1): 101. https://doi.org/10.1007/s40820-019-0332-7. |
| [56] | Luo D X, Lei P, Huang Y X, Tian G R, Xiang X D. Improved electrochemical performance of graphene-integrated NaTi2(PO4)3/C anode in high-concentration electrolyte for aqueous sodium-ion batteries[J]. J. Electroanal. Chem., 2019, 838: 66-72. https://doi.org/10.1016/j.jelechem.2019.02.057. |
| [57] | Han J, Zarrabeitia M, Mariani A, Jusys Z, Hekmatfar M, Zhang H, Geiger D, Kaiser U, Behm R J, Varzi A, Passerini S. Halide-free water-in-salt electrolytes for stable aqueous sodium-ion batteries[J]. Nano Energy, 2020, 77: 105176. https://doi.org/10.1016/j.nanoen.2020.105176. |
| [58] | Peng D D, Sun R T, Han J, Zhao T L, Tian R J, Zhang A R, Zhang Y C, You Y. A low-concentrated electrolyte with a 3.5 V electrochemical stability window, made by restructuring the H-bond network, for high-energy and long-life aqueous sodium-ion batteries[J]. ACS Energy Lett., 2024, 9(12): 6215-6224. https://doi.org/10.1021/acsenergylett.4c02901. |
| [59] | Liu T T, Wu H, Du X F, Wang J Z, Chen Z, Wang H, Sun J R, Zhang J J, Niu J P, Yao L S, Zhao J W, Cui G L. Water-locked eutectic electrolyte enables long-cycling aqueous sodium-ion batteries[J]. ACS Appl. Mater. Interfaces, 2022, 14(29): 33041-33051. https://doi.org/10.1021/acsami.2c04893. |
| [60] | Mohamed A I, Whitacre J F. Capacity fade of NaTi2(PO4)3 in aqueous electrolyte solutions: Relating pH increases to long term stability[J]. Electrochim. Acta, 2017, 235: 730-739. https://doi.org/10.1016/j.electacta.2017.03.106. |
| [61] | He Y T, Chen H Q, Wang Y J, Zhang Y M, Hou L R, Jiang R Y, Yuan C Z. Boosting sodium-storage behaviors of NASICON-type NaTi2(PO4)3 anode by synergistic modulations in both materials and electrolytes towards aqueous Na-ion batteries[J]. Electrochim. Acta, 2023, 447: 142128. https://doi.org/10.1016/j.electacta.2023.142128. |
| [62] | Kitajou A, Mitsuyasu T, Nagai T, Yoshida K, Kobayashi W. NaClO4 ethylene glycol-water binary solution as an electrolyte for aqueous sodium ion batteries[J]. Electrochemistry, 2023, 91(11): 117002. https://doi.org/10.5796/electrochemistry.23-00086. |
| [63] | Wang M J, Zhao J J, Zhang Y Y, Liu Y Y, Ji W, Xiang X D. Electrolyte‐salts regulated hydrogen‐bonding configuration and interphase formation achieving highly stable anode for rechargeable aqueous sodium‐ion batteries[J]. Small, 2024, 20(52): 2407961. https://doi.org/10.1002/smll.202407961. |
| [64] | Nian Q S, Liu S, Liu J, Zhang Q, Shi J, Liu C, Wang R, Tao Z L, Chen J. All-climate aqueous dual-ion hybrid battery with ultrahigh rate and ultralong life performance[J]. ACS Appl. Energy Mater., 2019, 2(6): 4370-4378. https://doi.org/10.1021/acsaem.9b00566. |
| [65] | Gou S Y, Zhang X Y, Xu Y H, Tang J H, Ji Y Y, Imran M, Pan L, Li J, Liu B-T. Inhibiting dissolution strategy achieving high-performance sodium titanium phosphate hybrid anode in seawater-based dual-ion battery[J]. J. Colloid Interface Sci., 2024, 675: 429-437. https://doi.org/10.1016/j.jcis.2024.07.022. |
| [66] | Zhang Q C, Man P, He B, Li C W, Li Q L, Pan Z H, Wang Z X, Yang J, Wang Z, Zhou Z Y, Lu X H, Niu Z Q, Yao Y, Wei L. Binder-free NaTi2(PO4)3 anodes for high-performance coaxial-fiber aqueous rechargeable sodium-ion batteries[J]. Nano Energy, 2020, 67: 104212. https://doi.org/10.1016/j.nanoen.2019.104212. |
| [67] | Zhao P, Yang X L, Liu Q C, Zhu J, Yang S D, Chen L, Zhang Q. Carbon-incorporated NaTi2(PO4)3on carbon cloth as a binder-free anode material for high-performance aqueous sodium-ion hybrid capacitors[J]. J. Porous Mater., 2024, 31(5): 1905-1913.10. https://doi.org/1007/s10934-024-01650-4. |
/
| 〈 |
|
〉 |