

离子液体增强质子转移并促进中性析氧反应
收稿日期: 2025-03-25
修回日期: 2025-04-30
录用日期: 2025-05-16
网络出版日期: 2025-05-16
Ionic Liquid Enhanced Proton Transfer for Neutral Oxygen Evolution Reaction
Received date: 2025-03-25
Revised date: 2025-04-30
Accepted date: 2025-05-16
Online published: 2025-05-16
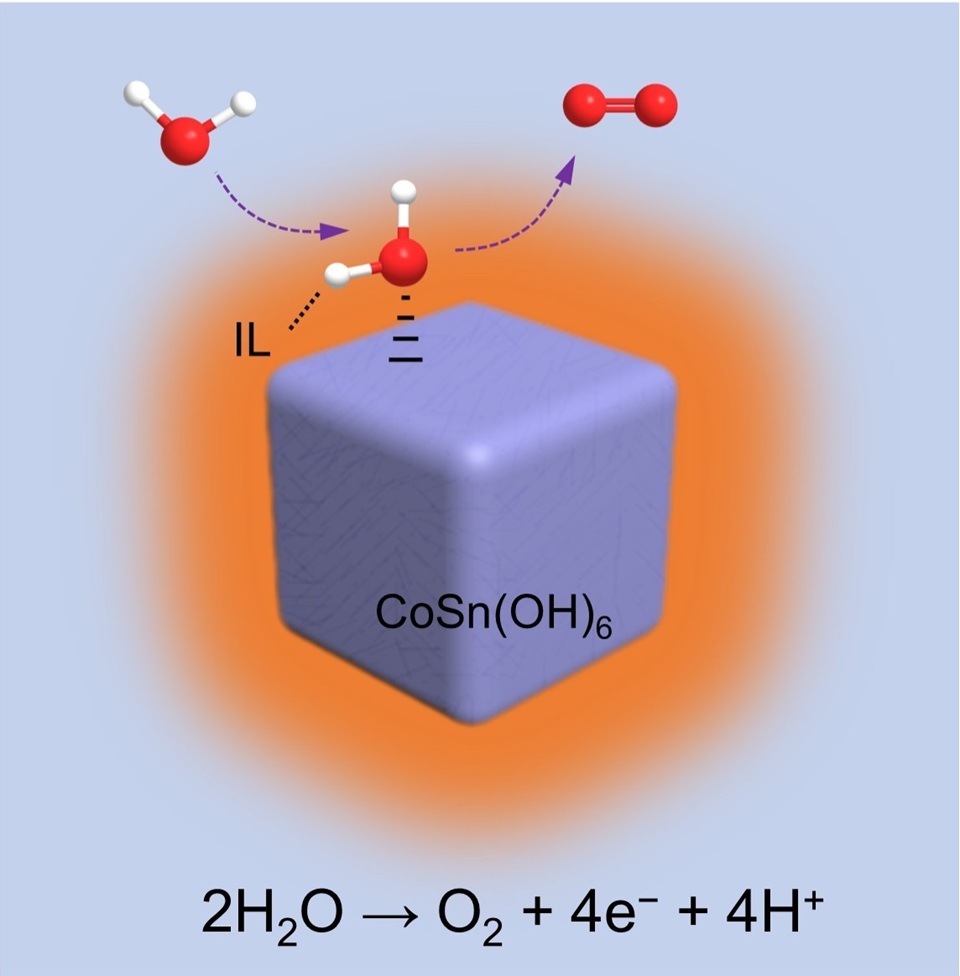
中性析氧反应对于新能源转换与存储领域而言意义十分重大。然而,缓慢的质子耦合电子转移步骤限制了析氧反应的整体效率。因此,开发高活性的中性析氧催化剂具有十分重要的研究价值。本文通过一种简便的策略成功制备出离子液体(IL)修饰的CoSn(OH)6纳米立方体(CoSn(OH)6-IL)。修饰的离子液体并未改变CoSn(OH)6的结构特征,但可有效调控活性位点附近的质子活性。与未经修饰的CoSn(OH)6纳米立方体相比较而言,CoSn(OH)6-IL对中性析氧反应展示出更高的本征活性。例如,在1.8 V(相对于可逆氢电极)的电压下,CoSn(OH)6-IL的析氧电流密度是CoSn(OH)6的4倍。根据pH依赖性实验、原位电化学阻抗图谱、化学探针实验和同位素效应等测试结果可以得出,CoSn(OH)6-IL催化剂中的界面离子液体可以作为质子转移介体,促进中性析氧反应的质子转移速率并提高析氧中间体的表面覆盖度,有效降低活化能垒,最终能够加速析氧反应动力学。这项工作提供了一种简单且有效的策略促进质子耦合电子转移过程,并有利于指导析氧催化剂的合理设计。
关键词: 电催化; 析氧反应; 离子液体; 质子转移; CoSn(OH)6立方体
陈明星 , 刘念 , 杜子翯 , 齐静 , 曹睿 . 离子液体增强质子转移并促进中性析氧反应[J]. 电化学, 2025 , 31(7) : 2515001 . DOI: 10.61558/2993-074X.3549
The development of highly active catalyst in pH-neutral media for oxygen evolution reaction (OER) is critical in the field of renewable energy storage and conversion. Nevertheless, the slow kinetics of proton-coupled electron transfer (PCET) hinders the overall OER efficiency. Herein, we report an ionic liquid (IL) modified CoSn(OH)6 nanocubes (denoted as CoSn(OH)6-IL), which could be prepared through a facile strategy. The modified IL would not change the structural characteristics of CoSn(OH)6, but could effectively regulate the local proton activity near the active sites. The CoSn(OH)6-IL exhibited higher intrinsic OER performances than the pristine CoSn(OH)6 in neutral media. For example, the current density of CoSn(OH)6-IL at 1.8 V versus reversible hydrogen electrode (RHE) was about 4 times higher than that of CoSn(OH)6. According to the pH-dependent kinetic investigations, operando electrochemical impedance spectroscopic and chemical probe tests, and deuterium kinetic isotope effects, the interfacial layer of IL could be utilized as a proton transfer mediator to promote the proton transfer, which enhances the surface coverage of OER intermediates and reduces the activation barrier. Consequently, the sluggish OER kinetics would be efficiently accelerated. This study provides a facile and effective strategy to facilitate the PCET processes and is beneficial to guide the rational design of OER electrocatalysts.

| [1] | Seh Z W, Kibsgaard J, Dickens C F, Chorkendorff I, Nørskov J K, Jaramillo T F. Combining theory and experiment in electrocatalysis: Insights into materials design[J]. Science, 2017, 355(6321): eaad4998. https://doi.org/10.1126/science.aad4998. |
| [2] | Qi J, Zhang W, Cao R. Solar-to-hydrogen energy conversion based on water splitting[J]. Adv. Energy Mater., 2018, 8(5): 1701620. https://doi.org/10.1002/aenm.201701620. |
| [3] | Wang T H, Tao L, Zhu X, Chen C, Chen W, Du S Q, Zhou Y Y, Zhou B, Wang D D, Xie C, Long P, Li W, Wang Y Y, Chen R, Zou Y Q, Wang S Y, Fu X Z, Li Y F, Duan X F. Combined anodic and cathodic hydrogen production from aldehyde oxidation and hydrogen evolution reaction[J]. Nat. Catal., 2022, 5(1): 66-73. https://doi.org/10.1038/s41929-021-00721-y. |
| [4] | Kumar A, Daw P, Milstein D. Homogeneous catalysis for sustainable energy: Hydrogen and methanol economies, fuels from biomass, and related topics[J]. Chem. Rev., 2022, 122(1): 385-441. https://doi.org/10.1021/acs.chemrev.1c00412. |
| [5] | Li W L, Li F S, Yang H, Wu X J, Zhang P L, Shan Y, Sun L C. A bio-inspired coordination polymer as outstanding water oxidation catalyst via second coordination sphere engineering[J]. Nat. Commun., 2019, 10(1): 5074. https://doi.org/10.1038/s41467-019-13052-1. |
| [6] | Zhang T, Liu Y P, Ye Q T, Fan H J. Alkaline seawater electrolysis at industrial level: Recent progress and perspective[J]. J. Electrochem., 2022, 28(10): 2214006. https://doi.org/10.13208/j.electrochem.2214006. |
| [7] | Zhang L S, Yuan H Y, Wang L P, Zhang H, Zang Y J, Tian Y, Wen Y Z, Ni F L, Song H, Wang H F, Zhang B, Peng H S. The critical role of electrochemically activated adsorbates in neutral OER[J]. Sci. China Mater., 2020, 63(12): 2509-2516. https://doi.org/10.1007/s40843-020-1390-6. |
| [8] | Qi J, Chen Q Z, Chen M X, Zhang W, Shen X X, Li J, Shangguan E, Cao R. Promoting oxygen evolution electrocatalysis by coordination engineering in cobalt phosphate[J]. Small, 2024, 20(38): 2403310. https://doi.org/10.1002/smll.202403310. |
| [9] | Takashima T, Ishikawa K, Irie H. Induction of concerted proton-coupled electron transfer during oxygen evolution on hematite using lanthanum oxide as a solid proton acceptor[J]. ACS Catal. 2019, 9(10): 9212-9215. https://doi.org/10.1021/acscatal.9b02936. |
| [10] | Gentry E C, Knowles R R. Synthetic applications of proton-coupled electron transfer[J]. Acc. Chem. Res. 2016, 49(8): 1546-1556. https://doi.org/10.1021/acs.accounts.6b00272. |
| [11] | Yamaguchi A, Inuzuka R, Takashima T, Hayashi T, Hashimoto K, Nakamura R. Regulating proton-coupled electron transfer for efficient water splitting by manganese oxides at neutral pH[J]. Nat. Commun., 2014, 5(1): 4256. https://doi.org/10.1038/ncomms5256. |
| [12] | Meyer T J, Huynh M H V, Thorp H H. The possible role of proton-coupled electron transfer (PCET) in water oxidation by photosystem II[J]. Angew. Chem. Int. Ed., 2007, 46(28): 5284-5304. https://doi.org/10.1002/anie.200600917. |
| [13] | Duan L K, Wang L, Li F, Li F S, Sun L C. Highly efficient bioinspired molecular Ru water oxidation catalysts with negatively charged backbone ligands[J]. Acc. Chem. Res., 2015, 48(7): 2084-2096. https://doi.org/10.1021/acs.accounts.5b00149. |
| [14] | Yano J, Yachandra V. Mn4Ca cluster in photosynthesis: Where and how water is oxidized to dioxygen[J]. Chem. Rev., 2014, 114(8): 4175-4205. https://doi.org/10.1021/cr4004874. |
| [15] | Lubitz W, Chrysina M, Cox N. Water oxidation in photosystem II[J]. Photosynth. Res., 2019, 142(1): 105-125. https://doi.org/10.1007/s11120-019-00648-3. |
| [16] | Lee Y V, Tian B. Learning from solar energy conversion: Biointerfaces for artificial photosynthesis and biological modulation[J]. Nano Lett., 2019, 19(4): 2189-2197. https://doi.org/10.1021/acs.nanolett.9b00388. |
| [17] | Gao X Q, Yang S J, Zhang W, Cao R. Biomimicking hydrogen-bonding network by ammoniated and hydrated manganese (II) phosphate for electrocatalytic water oxidation[J]. Acta Phys. Chim. Sin., 2021, 37(7): 2007031. https://doi.org/10.3866/pku.whxb202007031. |
| [18] | Liu F, Concepcion J J, Jurss J W, Cardolaccia T, Templeton J L, Meyer T J. Mechanisms of water oxidation from the blue dimer to photosystem II[J]. Inorg. Chem., 2008, 47(6): 1727-1752. https://doi.org/10.1021/ic701249s. |
| [19] | Siegbahn P E M. A structure-consistent mechanism for dioxygen formation in photosystem II[J]. Chem. Eur. J., 2008, 14(27): 8290-8302. https://doi.org/10.1002/chem.200800445. |
| [20] | Li W L, Li F S, Zhao Y L, Liu C, Li Y Z, Yang H, Fan K, Zhang P L, Shan Y, Sun L C. Promotion of the oxygen evolution performance of Ni-Fe layered hydroxides via the introduction of a proton-transfer mediator anion[J]. Sci. China Chem., 2022, 65(2): 382-390. https://doi.org/10.1007/s11426-021-1178-y. |
| [21] | Wang J W, Zhang X Q, Huang H H, Lu T B. A nickel(II) complex as a homogeneous electrocatalyst for water oxidation at neutral pH: Dual role of HPO42- in catalysis[J]. ChemCatChem, 2016, 8(20): 3287-3293. https://doi.org/10.1002/cctc.201600796. |
| [22] | Guo H B, Wang Y N, Guo K, Lei H T, Liang Z Z, Zhang X P, Cao R. A Co porphyrin with electron-withdrawing and hydrophilic substituents for improved electrocatalytic oxygen reduction[J]. J. Electrochem., 2022, 28(9): 2214002. https://doi.org/10.13208/j.electrochem.2214002. |
| [23] | Li R X, Zhao Y F, Liu Z M. Hydrogen bonding on ionic liquid reaction systems[J]. Sci. Sin.: Chim., 2022, 52(5): 655-667. https://doi.org/10.1360/ssc-2022-0012. |
| [24] | Dong K, Zhang S J, Wang D X, Yao X Q. Hydrogen bonds in imidazolium ionic liquids[J]. J. Phys. Chem. A, 2006, 110: 9775-9782. https://doi.org/10.1021/jp054054c. |
| [25] | Yu H Z, Bencherif S, Pham-Truong T N, Ghilane J. Immobilization of molecule-based ionic liquids: A promising approach to improve elecrocatalyst performance towards the hydrogen evolution reaction[J]. New J. Chem., 2022, 46(2): 454-464. https://doi.org/10.1039/D1NJ04400A. |
| [26] | Iijima G, Kitagawa T, Katayama A, Inomata T, Yamaguchi H, Suzuki K, Hirata K, Hijikata Y, Ito M, Masuda H. CO2 reduction promoted by imidazole supported on a phosphonium-type ionic-liquid-modified Au electrode at a low overpotential[J]. ACS Catal., 2018, 8(3): 1990-2000. https://doi.org/10.1021/acscatal.7b03274. |
| [27] | Wang T, Zhang Y, Huang B, Cai B, Rao R R, Giordano L, Sun S G, Shao-Horn Y. Enhancing oxygen reduction electrocatalysis by tuning interfacial hydrogen bonds[J]. Nat. Catal., 2021, 4(9): 753-762. https://doi.org/10.1038/s41929-021-00668-0. |
| [28] | Da-Yong S, Jing C. Hydrogen-bonding interactions between ionic liquid 1-ethyl-3-methylimidazolium trifluoromethanesulfonate and water[J]. Acta Phys. Chim. Sin., 2014, 30(9): 1605-1610. https://doi.org/10.3866/PKU.WHXB201407012 |
| [29] | Ji S, Li T, Gao Z D, Song Y Y, Xu J J. Boosting the oxygen evolution reaction performance of CoS2 microspheres by subtle ionic liquid modification[J]. Chem. Commun., 2018, 54(63): 8765-8768. https://doi.org/10.1039/C8CC05352A. |
| [30] | Koishi T. Molecular dynamics study of the effect of water on hydrophilic and hydrophobic ionic liquids[J]. J. Phys. Chem. B, 2018, 122(51): 12342-12350. https://doi.org/10.1021/acs.jpcb.8b07774. |
| [31] | Gao Y, Ye L, Cao S Y, Chen H, Yao Y N, Jiang J, Sun L C. Perovskite hydroxide CoSn(OH)6 nanocubes for efficient photoreduction of CO2 to CO[J]. ACS Sustainable Chem. Eng., 2018, 6(1): 781-786. https://doi.org/10.1021/acssuschemeng.7b03119. |
| [32] | Lin X H, Gao Y L, Jiang M, Zhang Y F, Hou Y D, Dai W X, Wang S B, Ding Z X. Photocatalytic CO2 reduction promoted by uniform perovskite hydroxide CoSn(OH)6 nanocubes[J]. Appl. Catal. B, 2018, 224: 1009-1016. https://doi.org/10.1016/j.apcatb.2017.11.035. |
| [33] | Paschoal V H, Faria L F O, Ribeiro M C C. Vibrational spectroscopy of ionic liquids[J]. Chem. Rev., 2017, 117(10): 7053-7112. https://doi.org/10.1021/acs.chemrev.6b00461. |
| [34] | Herstedt M, Smirnov M, Johansson P, Chami M, Grondin J, Servant L, Lassègues J C. Spectroscopic characterization of the conformational states of the bis(trifluoromethanesulfonyl)imide anion (TFSI-)[J]. J. Raman Spectrosc., 2005, 36(8): 762-770. https://doi.org/10.1002/jrs.1347. |
| [35] | Yang J, Liu H, Martens W N, Frost R L. Synthesis and characterization of cobalt hydroxide, cobalt oxyhydroxide, and cobalt oxide nanodiscs[J]. J. Phys. Chem. C, 2010, 114(1): 111-119. https://doi.org/10.1021/jp908548f. |
| [36] | Song F, Schenk K, Hu X. A nanoporous oxygen evolution catalyst synthesized by selective electrochemical etching of perovskite hydroxide CoSn(OH)6 nanocubes[J]. Energy Environ. Sci., 2016, 9(2): 473-477. https://doi.org/10.1039/C5EE03453A. |
| [37] | Chen M X, Li H J, Wu C L, Liang Y B, Qi J, Li J, Shangguan E, Zhang W, Cao R. Interfacial engineering of heterostructured Co(OH)2/NiPx nanosheets for enhanced oxygen evolution reaction[J]. Adv. Funct. Mater., 2022, 32(40): 2206407. https://doi.org/10.1002/adfm.202206407. |
| [38] | Satpathy B K, Raj C R, Pradhan D. Facile room temperature synthesis of CoSn(OH)6/g-C3N4 nanocomposite for oxygen evolution reaction[J]. Electrochim. Acta, 2022, 433: 141250. https://doi.org/10.1016/j.electacta.2022.141250. |
| [39] | Sahoo R, Sasmal A K, Ray C, Dutta S, Pal A, Pal T. Suitable morphology makes CoSn(OH)6 nanostructure a superior electrochemical pseudocapacitor[J]. ACS Appl. Mater. Interfaces, 2016, 8(28): 17987-17998. https://doi.org/10.1021/acsami.6b02568. |
| [40] | Lv D, Li Y, Wang L. Carbon aerogels derived from sodium lignin sulfonate embedded in carrageenan skeleton for methylene-blue removal[J]. Int. J. Biol. Macromol., 2020, 148: 979-987. https://doi.org/10.1016/j.ijbiomac.2020.01.136. |
| [41] | Li W, Li F, Zhao Y, Liu C, Li Y, Yang H, Fan K, Zhang P, Shan Y, Sun L. Promotion of the oxygen evolution performance of Ni-Fe layered hydroxides via the introduction of a proton-transfer mediator anion[J]. Sci. China Chem., 2022, 65(2): 382-390. https://doi.org/10.1007/s11426-021-1178-y. |
| [42] | Kurisaki T, Tanaka D, Inoue Y, Wakita H, Minofar B, Fukuda S, Ishiguro S-i, Umebayashi Y. Surface analysis of ionic liquids with and without lithium salt using X-ray photoelectron spectroscopy[J]. J. Phys. Chem. B, 2012, 116(35): 10870-10875. https://doi.org/10.1021/jp301658k. |
| [43] | Höfft O, Bahr S, Himmerlich M, Krischok S, Schaefer J A, Kempter V. Electronic structure of the surface of the ionic liquid [EMIM][Tf2N] studied by metastable impact electron spectroscopy (MIES), UPS, and XPS[J]. Langmuir, 2006, 22(17): 7120-7123. https://doi.org/10.1021/la060943v. |
| [44] | Smith E F, Rutten F J M, Villar-Garcia I J, Briggs D, Licence P. Ionic liquids in vacuo:? Analysis of liquid surfaces using ultra-high-vacuum techniques[J]. Langmuir, 2006, 22(22): 9386-9392. https://doi.org/10.1021/la061248q. |
| [45] | Zhang G R, Wolker T, Sandbeck D J S, Munoz M, Mayrhofer K J J, Cherevko S, Etzold B J M. Tuning the electrocatalytic performance of ionic liquid modified Pt catalysts for the oxygen reduction reaction via cationic chain engineering[J]. ACS Catal. 2018, 8(9): 8244-8254. https://doi.org/10.1021/acscatal.8b02018. |
| [46] | Freire M G, Carvalho P J, Gardas R L, Marrucho I M, Santos L M N B F, Coutinho J A P. Mutual solubilities of water and the [Cnmim][Tf2N] hydrophobic ionic liquids[J]. J. Phys. Chem. B, 2008, 112(6): 1604-1610. https://doi.org/10.1021/jp7097203. |
| [47] | Surendranath Y, Kanan M W, Nocera D G. Mechanistic studies of the oxygen evolution reaction by a cobalt-phosphate catalyst at neutral pH[J]. J. Am. Chem. Soc., 2010, 132(46): 16501-16509. https://doi.org/10.1021/ja106102b. |
| [48] | Ju M, Chen Z W, Zhu H, Cai R M, Lin Z D, Chen Y P, Wang Y J, Gao J L, Long X, Yang S H. Fe(III) docking-activated sites in layered birnessite for efficient water oxidation[J]. J. Am. Chem. Soc., 2023, 145(20): 11215-11226. https://doi.org/10.1021/jacs.3c01181. |
| [49] | Jin K, Park J, Lee J, Yang K D, Pradhan G K, Sim U, Jeong D, Jang H L, Park S, Kim D, Sung N-E, Kim S H, Han S, Nam K T. Hydrated manganese(II) phosphate (Mn3(PO4)2·3H2O) as a water oxidation catalyst[J]. J. Am. Chem. Soc., 2014, 136(20): 7435-7443. https://doi.org/10.1021/ja5026529. |
| [50] | Liu M, Li N, Wang X M, Zhao J, Zhong D C, Li W, Bu X H. Photosystem II inspired NiFe-based electrocatalysts for efficient water oxidation via second coordination sphere effect[J]. Angew. Chem. Int. Ed., 2023, 62(20): e202300507. https://doi.org/10.1002/anie.202300507. |
| [51] | Xiao Z H, Huang Y C, Dong C L, Xie C, Liu Z J, Du S Q, Chen W, Yan D F, Tao L, Shu Z W, Zhang G H, Duan H G, Wang Y Y, Zou Y Q, Chen R, Wang S Y. Operando identification of the dynamic behavior of oxygen vacancy-rich Co3O4 for oxygen evolution reaction[J]. J. Am. Chem. Soc., 2020, 142(28): 12087-12095. https://doi.org/10.1021/jacs.0c00257. |
| [52] | Zhou P, Lv X S, Tao S S, Wu J C, Wang H F, Wei X X, Wang T H, Zhou B, Lu Y X, Frauenheim T, Fu X, Z Wang S Y, Zou Y Q. Heterogeneous-interface-enhanced adsorption of organic and hydroxyl for biomass electrooxidation[J]. Adv. Mater., 2022, 34(42): 2204089. https://doi.org/10.1002/adma.202204089. |
| [53] | Zhao T W, Shen X J, Wang Y, Hocking R K, Li Y, Rong C L, Dastafkan K, Su Z, Zhao C. In situ reconstruction of V-doped Ni2P pre-catalysts with tunable electronic structures for water oxidation[J]. Adv. Funct. Mater., 2021, 31(25): 2100614. https://doi.org/10.1002/adfm.202100614. |
| [54] | Li L Q, Yang H B, Miao J W, Zhang L P, Wang H Y, Zeng Z P, Huang W, Dong X X, Liu B. Unraveling oxygen evolution reaction on carbon-based electrocatalysts: Effect of oxygen doping on adsorption of oxygenated intermediates[J]. ACS Energy Lett., 2017, 2(2): 294-300. https://doi.org/10.1021/acsenergylett.6b00681. |
| [55] | Tao H B, Xu Y H, Huang X, Chen J Z, Pei L J, Zhang J M, Chen J G, Liu B. A general method to probe oxygen evolution intermediates at operating conditions[J]. Joule, 2019, 3(6): 1498-1509. https://doi.org/10.1016/j.joule.2019.03.012. |
| [56] | Zhang B, Zheng X L, Voznyy O, Comin R, Bajdich M, García-Melchor M, Han L L, Xu J X, Liu M, Zheng L R, García de Arquer F P, Dinh C T, Fan F J, Yuan M J, Yassitepe E, Chen N, Regier T, Liu P F, Li Y H, De Luna P, Janmohamed A, Xin H L, Yang H G, Vojvodic A, Sargent E H. Homogeneously dispersed multimetal oxygen-evolving catalysts[J]. Science, 2016, 352(6283): 333-337. https://doi.org/10.1126/science.aaf1525. |
| [57] | Huang J Z, Sheng H Y, Ross R D, Han J, Wang X J, Song B, Jin S. Modifying redox properties and local bonding of Co3O4 by CeO2 enhances oxygen evolution catalysis in acid[J]. Nat. Commun., 2021, 12(1): 3036. https://doi.org/10.1038/s41467-021-23390-8. |
/
| 〈 |
|
〉 |