

在TiO2纳米阵列上电沉积RuO2用于CO2电还原
收稿日期: 2017-04-07
修回日期: 2017-04-28
网络出版日期: 2017-04-28
基金资助
This work is supported by the 973 Program (No. 2015CB932303) of MOST and NSFC (No. 21473039).
Electrodeposition of RuO2 Layers on TiO2 Nanotube Array toward CO2 Electroreduction
Received date: 2017-04-07
Revised date: 2017-04-28
Online published: 2017-04-28
Supported by
This work is supported by the 973 Program (No. 2015CB932303) of MOST and NSFC (No. 21473039).
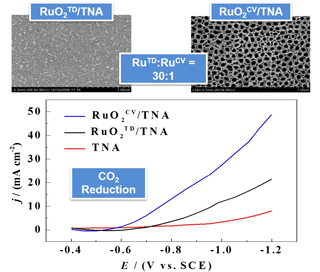
传统上,RuO2/TiO2复合电极制备是通过在TiO2/Ti基体上多次涂覆含Ru前驱体溶液和随后热分解(TD)来实现的. 为克服上述方法中Ru用量大和利用率低之不足, 本工作主要基于循环伏安法(CV)在TiO2纳米管阵列(TNA)上电沉积RuO2制备RuO2CV/TNA复合电极. SEM、GIXRD和CV结果表明, 电沉积的RuO2为无定型结构, 所制备电极中的Ru用量约为传统的RuO2TD/TNA电极中Ru用量的1/30. 尽管两电极催化CO2还原产物的法拉第效率接近, 但是RuO2CV/TNA电极比RuO2TD/TNA电极展示了更高的还原电流, 较正的初始还原电位和更好的稳定性. 与磷酸盐缓冲溶液中电还原CO2相比,RuO2CV/TNA电极在0.1 mol•L-1 KHCO3中电还原CO2除生成更高法拉第效率的甲酸根和甲烷外,还检测到CO的生成.
蒋 孛 , 张莉娜 , 秦先贤 , 蔡文斌 . 在TiO2纳米阵列上电沉积RuO2用于CO2电还原[J]. 电化学, 2017 , 23(2) : 238 -244 . DOI: 10.13208/j.electrochem.161253
RuO2/TiO2 composite materials have multitude of electrocatalytic applications including but not limited to CO2 reduction reaction (CO2RR). RuO2/TiO2 electrodes were previously prepared by repetitive coating and thermal decomposition (TD) of a Ru(III) precursor solution on Ti substrate. In this work, electrochemical potential cycling is applied to deposit amorphous RuO2 (α-RuO2) layers onto TiO2 nanotube array (TNA) (RuO2CV/TNA) preformed on Ti foil. SEM, GIXRD, and voltammetry are applied to characterize the structures of the resulting RuO2CV/TNA. Ru loading on the RuO2CV/TNA electrode is ca. 1/30 of that on the conventional RuO2TD/TNA electrode. Although both electrodes yield similar faradaic efficiencies (FEs) for the reduction products, the RuO2CV/TNA electrode displays a much higher reduction current, a more positive initial reduction potential and a better durability than the RuO2TD/TNA one. In addition to higher FEs for formate and CH4, the RuO2CV/TNA electrode yields the product of CO for the CO2RR in 0.1 mo•lL-1 KHCO3, which is not available in a PBS solution with pH 7.

Key words: CO2 reduction; amorphous RuO2; TiO2 nanotube array; electrodeposition
/
| 〈 |
|
〉 |