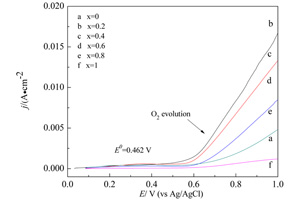
用热分解法制得不同混合比例的二元金属氧化物催化剂CoxCr1-xO3/2(x = 0,0.2,0.4,0.6,0.8,1),使用SEM、XRD和XPS观察表征催化剂形貌、晶型和价态,使用线性扫描伏安、阶梯波伏安和恒电位测试电极活性、过电位和稳定性. 结果表明,该Co3O4和绿铬矿型Cr2O3混合物形成固溶体CoxCr1-xO3/2. x = 0.2时,Co0.2Cr0.8O3/2电极性能较单一Co3O4和Cr2O3电极好,在高电位(1.0 V vs. Ag/AgCl),其电流强度是Co3O4的3.75倍,Cr2O3的15.2倍,其过电位(η = 0.0703 V)也较Co3O4(η = 0.6109 V)和Cr2O3(η = 0.435 V)小,催化性能最好,在强碱性溶液(pH=13)中有良好的稳定性.
Binary metal mixed oxides of Co and Cr with nominal compositional formulas, CoxCr1-xO3/2 (x=0, 0.2, 0.4, 0.6, 0.8 and 1.0), were obtained by thermal decomposition method. The morphologies, crystal forms and valence states of the catalyst powders were analyzed by SEM, XRD and XPS, respectively. And the electrochemical properties, overpotentials and stabilities of CoxCr1-xO3/2 electrodes for oxygen evolution reaction (OER) in an alkaline solution were evaluated by linear sweep voltammetry (LSV), staircase voltammetry and chronoamperometry, respectively. The results indicated that the mixtures of Co3O4 and eskolaite-Cr2O3 formed solid solutions of CoxCr1-xO3/2. When x=0.2, the electrocatalytic performance of Co0.2Cr0.8O3/2 catalyst was better than those of Co3O4 and Cr2O3. At high potential region (1.0 V vs. Ag/AgCl), the current density of Co0.2Cr0.8O3/2 catalyst was 3.75 times higher than that of Co3O4 and 15.2 times higher than that of Cr2O3. Furthermore, the overpotential of Co0.2Cr0.8O3/2 catalyst (η = 0.0703 V at a current density of 1×10-3 A·cm-2) was much lower than those of Co3O4 (η = 0.6109 V) and Cr2O3 (η = 0.4350 V), showing the best electrocatalytic property. In addition, the Co0.2Cr0.8O3/2 catalyst had good stability in strong alkaline solution (pH = 13) during OER process.