

基于DFT计算的Pt(111)表面氧覆盖度和水合质子模型对氧还原反应路径的影响(英文)
收稿日期: 2014-03-19
修回日期: 2014-05-02
网络出版日期: 2014-05-09
基金资助
This work was supported by Ministry of Science and Technology (Grant Nos. 2012CB932800 and 2013AA110201), the National Natural Science Foundation of China (Grant No. 21303048), Hunan Provincial Natural Science Foundation of China (Grant No. 13JJ4101), and Foundation of the Education Department of Hunan Province (Grant No. 12C0832)
Effects of Oxygen Coverage and Hydrated Proton Model on the DFT Calculation of Oxygen Reduction Pathways on the Pt(111) Surface
Received date: 2014-03-19
Revised date: 2014-05-02
Online published: 2014-05-09
Supported by
This work was supported by Ministry of Science and Technology (Grant Nos. 2012CB932800 and 2013AA110201), the National Natural Science Foundation of China (Grant No. 21303048), Hunan Provincial Natural Science Foundation of China (Grant No. 13JJ4101), and Foundation of the Education Department of Hunan Province (Grant No. 12C0832)
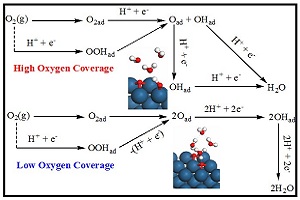
欧利辉 , 陈胜利 . 基于DFT计算的Pt(111)表面氧覆盖度和水合质子模型对氧还原反应路径的影响(英文)[J]. 电化学, 2014 , 20(3) : 206 -218 . DOI: 10.13208/j.electrochem.130894

Key words: minimum energy path; ORR mechanism; oxygen coverage; hydrated proton model
/
| 〈 |
|
〉 |