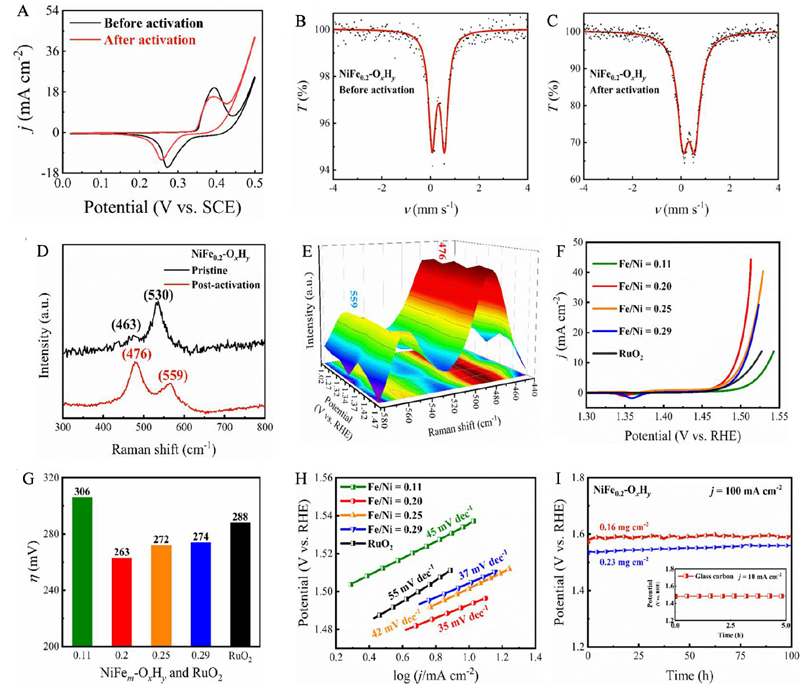
Figure 14. (A) Cyclic voltammetric curves of NiFe0.2-OxHy before (the black curves, α-phase Ni(OH)2 structure) and after electrochemical activation (the red curves, γ-phase NiOOH structure). (B-C) 57Fe Mössbauer spectra of NiFe0.2-OxHy before and after electrochemical activation. (D) Raman spectra of NiFe0.2-OxHy before (black) and after (red) applying anodic potential. (E) Operando Raman spectra of NiFe0.2-OxHy collected at different applied potentials (V vs. RHE). (F) The OER polarization curves with iR correction. (G) Overpotentials at 10 mA·cm-2. (H) Tafel plots of NiFem-OxHy with different molar ratios of Fe/Ni and commercial RuO2. (I) Chronopotentiometric curves of NiFe0.2-OxHy on Ni foam with different catalyst loadings at a constant current density of 100 mA·cm-2 for 100 h. The inset shows the chronopotentiometric curve of NiFe0.2-OxHy at a constant current density of 10 mA·cm-2[
.